Abstract
Objective: Due to the negative connotations around radiation, there is a great deal of angst in the community regarding radiation exposure and health; especially electromagnetic radiation (EMR) sources such as powerlines, mobile phone towers and the rollout of the 5G network. As such, it is important for health authorities to provide the public with information and assurances regarding radiation safety. The Australian Radiation Protection and Nuclear Safety Agency (ARPANSA) set up community engagement programs to address community concerns.
Type of program or service: From 2003 until April 2022, ARPANSA operated a Health Complaints Register, which collected reports of health complaints from members of the public related to possible EMR exposures.
Methods: Collected data was used to produce annual statistical summaries on the nature and level of complaints received. Since 2016, ARPANSA has also run the Talk to a Scientist program, which allows the public to communicate directly with scientists on issues about radiation exposure, health and protection in Australia. Data is collected on the type of radiation and radiation source.
Results: There was a low level of interest in the Register, with only 180 reports received over the duration of its operation. Smart meters were the most common source of EMR exposure reported to be responsible for adverse health effects. The most common adverse health effect reported was headaches. The Register was closed in April 2022 due to a lack of interest. In contrast, the Talk to a Scientist program has responded to 6546 enquiries since 2016, most of which have been on EMR sources and the success of the Talk to a Scientist program, which rendered the Register obsolete.
Lessons learnt: The EMR Health Complaints Register never received much interest from the public, potentially due to a perceived lack of engagement with authorities. The Talk to a Scientist program, which facilitated direct interaction with subject matter experts, has been much more successful in engaging with the public and addressing community concerns on radiation safety.
Full text
Background
The Australian population is constantly exposed to radiation from various natural and artificial sources. Due to the negative connotations around radiation, there is a great deal of angst in the community regarding radiation exposure and health. This has been especially evident in Australia with electromagnetic radiation (EMR) sources such as powerlines, mobile phone towers and, more recently, the 5G network rollout. Further, some members of the community have reported experiencing health problems, such as headaches, dizziness and fatigue which they attribute to EMR radiation exposure.1 Some claim to have electromagnetic hypersensitivity (EHS), a condition in which non-specific symptoms are attributed to EMR exposure.2,3 As such, it is important for health authorities to provide the public with information and assurances regarding the safety of EMR radiation as well as other radiation sources such as ionising radiation, ultraviolet (UV) radiation, and other optical sources.
The Australian Radiation Protection and Nuclear Safety Agency (ARPANSA) is the Australian Government’s primary authority on radiation protection and nuclear safety. ARPANSA protects the Australian people and the environment from the harmful effects of radiation through research, regulation and engagement with the community. ARPANSA established the Australian EMR Health Complaints Register (the Register) in 2003, following a recommendation by the Australian Senate Inquiry into EMR in 2000, to formulate a centralised complaints mechanism for people to report adverse health effects associated with mobile phone use and other EMR radiation exposure sources.4 The purpose of the Register was to gather information that ARPANSA could use to research the health effects of EMR exposure on people and the environment.
Since 2016, ARPANSA has also run the Talk to a Scientist (TTAS) program, a key forum in which ARPANSA engages with the public, helping to alleviate concern and deliver its mission for a safe radiation environment in Australia. This program provides the public with the opportunity to communicate directly with ARPANSA scientists on radiation exposure, health, and protection issues. The TTAS program has been at the forefront of ARPANSA’s communications activities and has evolved into a fully integrated element of the agency’s broader community engagement strategy.
Methods
The EMR Health Complaints Register
Members of the public who believed they have suffered adverse health effects as a result of exposure to EMR could lodge a written complaint to the Register using a standardised form that gathered data on demographics, the symptoms believed to be caused by EMR exposure (e.g. headache, dizziness, fatigue) and the source of EMR exposure believed to be causing symptoms (e.g. mobile phone tower, powerline, wi-fi). The Register operated in strict compliance with the Privacy Act 1988 and, as such, personal information on people reporting may not be disclosed. ARPANSA did not investigate or attempt to resolve individual complaints. However, the information was used to produce annual summaries on the number and nature of reports received. Data were collected from July 2003 until the cessation of the Register in April 2022. Details regarding the Register were available on the ARPANSA website and were advertised by other government agencies. The Register was also promoted in various forums, including community groups and other stakeholders.
The Talk To A Scientist program
Enquiries on any radiation topic are received by ARPANSA via phone or email and are responded to by a dedicated team of ARPANSA scientists. Phone lines are open to the general public on Tuesdays and Thursdays from 11:00 am to 12:30 pm AEST, excluding public holidays, while email enquiries can be received any time. The TTAS program aims to respond to all email enquiries within five business days; however, some emails take longer. All email and call enquiries are logged, and data is collected on the type of radiation (e.g. EMR radiation, ionising radiation, UV radiation) and the specific radiation source (e.g. mobile phone base station, powerline, 5G). This data provides ARPANSA valuable insight into community concerns and risk perception trends. Data has been collected since the beginning of the TTAS program in 2016. The TTAS program is advertised on the ARPANSA website and by other government agencies and non-government organisations with a stake in radiation protection. More recently, the TTAS program is also advertised on ARPANSA social media channels.
Results
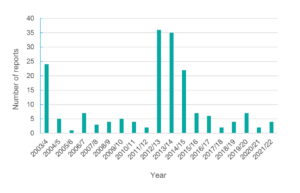
The EMR Health Complaints Register has only received 180 health complaint reports since it started. The number of reports received for different years is shown in Figure 1. The peak reporting occurred in the 2012–13 and 2013–14 financial years when 36 and 35 complaints were reported, respectively, primarily concerning smart meters, which were introduced in Australia at that time. However, in the last 6 years, an average of only 4-5 health complaints were received per year, with the 2020–21 financial year recording only two health complaints. This spans the period of the introduction of the 5G mobile phone network, which did not see a considerable increase in reporting. The most common EMR radiation sources and health complaints reported are shown in Table 1. Smart meters were the most common EMR source (n = 71), while headaches were the most common health complaint (n = 92), noting that some reports included more than one EMR source and more than one health complaint. There were only three health complaints attributed to a 5G source. Due to the low number of reports received and the expansion of the Agency’s TTAS program, ARPANSA closed the Australian EMR Health Complaints Register in April 2022.
Figure 1. Number of electromagnetic radiation (EMR) health complaints made to the EMR Health Complaints Register by yeara (click on figure to enlarge)
a EMR Health Complaints Register reported data based on financial year
Table 1a. Top five sources of electromagnetic radiation (EMR) reported to the EMR Health Complaints Register from July 2003 to April 2022
| EMR source | Reportsa |
| Smart meter | 71 |
| Base station | 48 |
| Mobile phone | 47 |
| Wi-fi | 37 |
| Household electrical appliances | 29 |
aSome reports included more than one EMR source and more than one health complaint.
Table 1b. Top five adverse health effects reported to the EMR Health Complaints Register from July 2003 to April 2022
| Reported health effects | Reportsa |
| Headaches | 92 |
| Anxiety | 54 |
| Body pain | 53 |
| Disturbed sleep | 52 |
| Lethargy | 49 |
aSome reports included more than one EMR source and more than one health complaint.
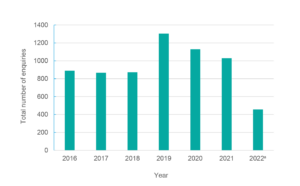
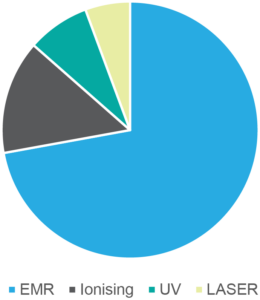
The TTAS Program has responded to 6546 enquiries from its launch in 2016 to 4 August 2022; the number of enquiries per year are shown in Figure 2. Most enquiries have been on EMR radiation, accounting for 65% of all questions as shown in Figure 3. Ionising radiation and UV account for 13% and 7% of the enquiries, respectively. Electricity supply infrastructure (which includes powerlines, substations and transformers) and telecommunications infrastructure (mobile phone base stations and broadcast towers) have consistently been the most frequent radiation sources about which enquires have been made. The number of enquiries significantly increased in 2019, as shown in Figure 3, largely due to the 5G rollout, which accounted for 25% of all enquiries that year. The number of queries has been falling since then, primarily due to a decrease in 5G enquiries, and are expected to return to the levels seen in 2016–2018.
Figure 2. Number of Talk to a Scientist (TTAS) enquiries per yeara (click on figure to enlarge)
Figure 3. Number of Talk to a Scientist (TTAS) enquiries since 2016 by radiation type (click on figure to enlarge)
EMR=electromagnetic radiation; Ionising=ionising radiation; UV=ultraviolet radation; Laser=laser radiation.
Discussion
Apart from four separate years in which a moderate number of complaints were received (as shown in Figure 1), the EMR Health Complaints Register did not attract significant interest from the public in its 18 years of operation. Less than 10 health complaints were recorded in each of 14 years of its operation. The low level of interest was especially evident in the last few years of operation, where increased public concern over the 5G network rollout did not transpire in a rise in Register reporting. The low number of reports to the Register was likely due to the lack of engagement with complainants, with feedback provided to ARPANSA by complainants to that effect. Although the complainant received confirmation that their health complaint had been received and information recorded, there was no risk communication on the health risks of EMR-emitting devices or further information that may contextualise the symptoms they were experiencing. This is important to note, as EHS has been attributed to psychological mechanisms such as the ‘nocebo effect’ (in which a negative outcome occurs due to a belief that a factor can cause harm).2,5-8 Without providing effective risk communication to inform risk perceptions, these symptoms are unlikely to improve and may worsen.
The decline in reporting to the EMR Health Complaints Register also coincided with the introduction of the TTAS program, which began in 2016. Unlike the Register, the TTAS program provides the general public with the opportunity to discuss concerns and questions relating to EMR exposure as well as other types of radiation and receive advice and information directly from ARPANSA scientists. This facilitates ‘two-way’ communication between scientists and the general public, unlike the Register, which was often perceived as a ‘one-way’ process. Further, the TTAS program directs people, especially individuals suffering from EHS, to medical professionals or other relevant authorities. In 2020, the TTAS program responded to 859 enquiries on EMR radiation alone. This included a significant increase in 5G enquires, which accounted for 441 of those enquiries, whereas in comparison, the EMR Health Complaints Register only recorded three health complaints attributed to 5G during the duration of its active operation. The TTAS program addresses a broader range of community concerns, not just health complaints, which partially explains the significantly higher number of enquiries received by TTAS compared to the EMR Health Complaints Register. However, many TTAS enquiries are from members of the general public reporting health complaints. In these instances, scientists provide general health advice but cannot, and do not, assess individual symptoms or diagnose health conditions. Those seeking specific medical advice are advised to see a medical professional to discuss their symptoms or conditions. ARPANSA has not formally evaluated the level of satisfaction of people contacting TTAS and whether their concerns have been addressed, but such an investigation will form future improvements to the program.
The data gathered from the TTAS program indicates that EMR radiation is of far greater concern to the community than ionising, UV or other types of radiation, with electricity supply infrastructure and telecommunications infrastructure being the most common types of enquiries. This may be due to the visible nature of this infrastructure around the community and the misinformation available online regarding possible health effects. The data also shows that new technologies like 5G increase public concern initially, but the anxiety tends to subside as people get used to the technology. It is important to highlight that public exposure to EMR is extremely low9,10, and there is no substantiated evidence of harm as opposed to the harmful effects of UV radiation from the sun, noting that Australia has one of the highest rates of skin cancer in the world.11
A person’s emotions and beliefs often influence risk perception.12 This has been demonstrated in previous research on EMR, particularly regarding the 5G mobile network, which has found that high levels of perceived exposure, mistrust in institutions, and dread, as well as evaluation of public policies as being ineffective, are associated with increased risk perception.13-15 This can often lead to sudden judgements about risk, as opposed to a more scientific analysis which would lead to a different assessment.16 A recent systematic review on EMR risk communication recommended that risk communicators explain exposure patterns more thoroughly, as they found that after receiving explanations about exposure patterns, recipients adjusted their risk perception.17 This review also found that most risk communication was textual, which can limit the effectiveness of the communication. These findings support the open dialogue facilitated by the TTAS call program for effectively communicating risk. Therefore, it is essential to continue engaging with the public to reduce community concern and also to raise awareness of the real risks of radiation so the public can make more informed decisions on how they manage such risks.18
Conclusion and lessons learnt
The EMR Health Complaints Register was operational for 18 years, but the annual statistical summaries produced were limited due to the low level of engagement. The absence of interest in the Register may be due to the perceived lack of attention from authorities and lack of follow-up when making a health complaint. The TTAS program has been far more effective in ARPANSA’s community engagement activities. This is likely due to the direct engagement with subject matter experts, which has received far greater interest from the public and allows ARPANSA to communicate risks more effectively. Data gathered from the TTAS program has also shown that sections of the general public can fear new technological infrastructure. However, these concerns subside as the technology becomes more mainstream, as with 5G. The direct correspondence via the TTAS program is a significant improvement on the Register, as it offers greater opportunity to address psychological mechanisms which are the most likely cause of reported health symptoms. ARPANSA can more effectively inform risk perception and reduce community concerns by improving risk communication.
Acknowledgements
The authors would like to acknowledge Lydiawati Tjong and Rohan Mate for assisting with managing the EMR Health Complaints Register and Samantha Gunther for archiving complaint records. We also acknowledge all past and current ARPANSA scientists who have engaged with members of the public via the TTAS program.
Peer review and provenance
Externally peer reviewed, not commissioned.
Copyright:
© 2023 Brzozek and Karipidis. This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International Licence, which allows others to redistribute, adapt and share this work non-commercially provided they attribute the work and any adapted version of it is distributed under the same Creative Commons licence terms.
References
- 1. Eltiti S, Wallace D, Russo R, Fox E. Symptom presentation in idiopathic environmental intolerance with attribution to electromagnetic fields: evidence for a nocebo effect based on data re-analyzed from two previous provocation studies. Front Psychol. 2018;9:1563. CrossRef | PubMed
- 2. Dieudonné M. Electromagnetic hypersensitivity: a critical review of explanatory hypotheses. Environ Health. 2020;19(1):48. CrossRef | PubMed
- 3. Seitz H, Stinner D, Eikmann T, Herr C, Röösli M. Electromagnetic hypersensitivity (EHS) and subjective health complaints associated with electromagnetic fields of mobile phone communication – a literature review published between 2000 and 2004. Sci. Total Environ. 2005;349(1-3):45–55. CrossRef | PubMed
- 4. Commonwealth of Australia. Inquiry into Electromagnetic Radiation. Canberra: Australian Parliament; 2001 [cited 2023 Jul 11]. Available from:www.aph.gov.au/Parliamentary_Business/Committees/Senate/Environment_and_Communications/Completed_inquiries/1999-02/emr/report/index
- 5. Berthelot JM. Is electromagnetic hypersensitivity entirely ascribable to nocebo effects? Joint Bone Spine. 2016;83(2):121–3. CrossRef | PubMed
- 6. Bräscher AK, Schulz SM, Van den Bergh O, Witthöft M. Prospective study of nocebo effects related to symptoms of idiopathic environmental intolerance attributed to electromagnetic fields (IEI-EMF). Environ Res. 2020;190:110019. CrossRef | PubMed
- 7. Dieudonné M. Does electromagnetic hypersensitivity originate from nocebo responses? Indications from a qualitative study. Bioelectromagnetics. 2016;37(1):14–24. CrossRef | PubMed
- 8. Verrender A, Loughran SP, Dalecki A, Freudenstein F, Croft RJ. Can explicit suggestions about the harmfulness of EMF exposure exacerbate a nocebo response in healthy controls? Environ Res. 2018;166:409–17. CrossRef | PubMed
- 9. Karipidis K, Henderson S, Wijayasinghe D, Tjong L, Tinker R. Exposure to radiofrequency electromagnetic fields from wi-fi in Australian schools. Radiat Prot Dosimetry. 2017;175(4):432–9. CrossRef | PubMed
- 10. Karipidis KK. Survey of residential power-frequency magnetic fields in Melbourne, Australia. Radiat Prot Dosimetry. 2015;163(1):81–91. CrossRef | PubMed
- 11. Walker H, Maitland C, Tabbakh T, Preston P, Wakefield M, Sinclair C. Forty years of Slip! Slop! Slap! A call to action on skin cancer prevention for Australia. Public Health Res Pract. 2022;32(1):e31452117. CrossRef | PubMed
- 12. Thomas SP, Hrudey S, Hrudey SE. Risk of death in Canada: What we know and how we know it. Canada, University of Alberta Press, 1997.
- 13. Koh TH, Choi JW, Seo M, Choi HD, Kim K. Factors affecting risk perception of electromagnetic waves from 5G network base stations. Bioelectromagnetics. 2020;41(7):491–9. CrossRef | PubMed
- 14. Frey R. Psychological drivers of individual differences in risk perception: a systematic case study focusing on 5G. Psychol Sci. 2021;32(10):1592–604. CrossRef | PubMed
- 15. Freudenstein F, Wiedemann PM, Brown TW. Exposure perception as a key indicator of risk perception and acceptance of sources of radio frequency electromagnetic fields. J Environ Public Health. 2015;2015:198272. CrossRef | PubMed
- 16. Finucane ML. The psychology of risk judgements and decisions. In: Cromar N. Environmental Health in Australia and New Zealand. Oxford University Press, 2004.
- 17. Boehmert C, Freudenstein F, Wiedemann P. A systematic review of health risk communication about EMFs from wireless technologies. J Risk Res. 2020 May 3;23(5):571–97. CrossRef
- 18. Reckelhoff-Dangel C, Petersen DD. Risk communication in action: the risk communication workbook. Cincinnati, OH: United States Environmental Protection Agency, Office of Research and Development, National Risk Management Research Laboratory; 2007 [cited 2023 Jul 18]. Available from: www.epa.gov/sites/default/files/2020-12/documents/risk-communication-risk-communication-workbook.pdf