Abstract
Objectives: Despite the widely publicised health benefits of participation in bowel cancer screening, only 43.5% of recipients participate in the Australian National Bowel Cancer Screening Program (NBCSP). Through consultation with kit recipients, this study aimed to identify features of home bowel screening kits that could be modified to increase their use.
Method: Participants (n = 25) were presented with nine different bowel cancer screening kits and asked to identify features of each kit that might prevent or promote their use. Responses were coded using content analysis, and a narrative synthesis is presented summarising preferences relating to each element of the kit.
Results: Six modifiable elements were identified: collection tool, collection sheet, specimen container, instruction, packaging and processes. Participant preferences were for collection devices that limited the users’ proximity to faecal matter, smaller packaging, simpler processes and step-by-step pictorial instructions. Responses regarding aesthetics, the amount of information included and receiving immediate results were mixed.
Conclusions: Findings provide several consumer-driven recommendations, which are to be tested in future research aimed at improving the acceptability and usability of kits distributed in population bowel cancer screening programs.
Full text
Background
Colorectal cancer (CRC) is the third most common cancer and third largest cause of cancer death worldwide.1 More than 5000 Australians die from CRC each year and it is the second leading cause of cancer-related deaths in the country.2 If CRC is detected early, before symptoms become apparent, 5-year relative survival rates are as high as 93%.3 In Australia, like in many international settings, the Federal Government provides a population-wide National Bowel Cancer Screening Program (NBCSP) whereby all adults aged between 50 and 74 years are delivered a home bowel cancer screening test kit in the mail every 2 years. Such programs have the potential to substantially reduce CRC mortality and incidence, and reduce treatment costs.4 However, participation rates in such programs are low worldwide5, limiting their potential effectiveness. In Australia, only 43.5% of participants complete and return their NBCSP home test kit.6 Increasing these rates to just 60% would save an extra 25 000 lives and A$2 billion in healthcare costs by 2040.7
The home faecal occult blood test (FOBT) for bowel cancer screening that is currently distributed in the Australian NBCSP requires the user to insert a 6 cm plastic collection tool into the stool several times, then place the collection tool with the sample into a plastic test tube. The handle of the collection tool is then twisted on, becoming the lid of the test tube. Test tubes containing two samples are then placed in a two-compartment padded pouch and inside a zip-lock plastic bag to be stored in the refrigerator until the user is ready to mail them to the pathology lab in a padded pre-addressed envelope that is provided with the kit (see Kit 1, Supplementary File 1, available from: figshare.com/articles/online_resource/Supplementary_File_1_Goodwin_et_al_2022/19388600).
A recent scale development study identified and quantified several key barriers to bowel cancer screening.8 Psychological and attitudinal barriers such as fear and concerns around autonomy were evident, however, practical and physical barriers to kit use such as misplacing the test kit, hygiene concerns and physical challenges associated with collecting and storing a stool sample were more commonly reported.8 Overcoming these barriers to kit use is vital. Findings of a recent systematic review and meta-analysis of interventions to increase participation in home bowel cancer screening showed that simplifying the testing procedure can increase participation by up to 7%.9 Also, interventions that employ behavioural change techniques targeting physical aspects of the kit (e.g., reducing the amount of samples the user needs to collect) are more likely to be effective than those targeting contextual factors of home bowel cancer screening such as offering financial incentives or reducing negative emotions related to cancer screening.10 These results suggest that improving the useability of the kit itself should be a focus in efforts towards increasing participation rates.
According to the principles of human-centric design, a key step in improving user experience in any context is to identify and address the functional constraints associated with a task through consultation with end users.11 Previous studies have successfully employed this strategy to inform improvements to collection processes for HIV self-test kits12, instructions for test kits targeting cholesterol13, and home bowel cancer screening.14
Study Aims
Although research broadly suggests that physical changes to home test kits are likely to lead to increased use9,15, we are not aware of any studies to date that have consulted consumers directly to identify exact features of test kits that should be modified. This study aims to identify, through consultation with end users, the physical features of home bowel screening kits that exist or could be modified to facilitate ease of use. Findings will inform recommendations for changes to kit design that may improve home bowel cancer screening participation in population mail-out programs.
Methods
Participants
Participants (n = 25) were NBCSP recipients recruited through several mechanisms including an online survey about bowel cancer screening8 as well as invitations to professional, community and personal networks through social media and email. Examples of groups approached include local community noticeboard Facebook groups, bowling, golf and surf clubs in Queensland, Australia, and university staff forums. Adults aged between 50 and 74 years of age, living in Australia, were eligible to take part in the study. The sample consisted of 11 males and 14 females, 12 of whom had used the current NBCSP kit and 13 who had not used it (10 participants had never used any bowel cancer screening kit). Participant ages ranged from 51 to 72 (mean age 59.00; standard deviation [SD] 5.59). Recruitment ceased when data saturation was reached (as described below). Participants provided written informed consent and received an A$50 grocery voucher for participation. The study received approval from the University of Southern Queensland Human Research Ethics Committee (HI9REA291).
Materials
One current NBCSP home FOBT kit was provided to researchers by the program organisers. This kit requires the user to collect two stool samples from two separate bowel motions with the two sets of test tubes and collection tools provided. Two stool collection sheets are provided for the user to put into the toilet bowl to catch their bowel movement before it reaches the toilet water. A two-compartment padded pouch is provided to deposit each completed test. This pouch goes inside a zip-lock bag with a single opening that the user is advised to keep in the refrigerator until they are ready to post it to the laboratory for testing in the padded, pre-addressed reply-paid envelope.
A further eight home FOBT kits were sourced from Australia and internationally by contacting suppliers and purchasing through online shopping websites. This included one kit that had been used previously in the NBCSP (kit number 8). The variance in existing kit types were well represented in terms of: 1) packaging and contents including stool collection devices and containers; and 2) instructions and processes. For example, testing kits that allowed the user to immediately see their results and those which required testing in a pathology laboratory were included. Stool collection devices within the various kits included plastic spatulas with grooved tips, flat wooden spatulas and a brush. Where included, toilet liners provided with kits also varied in shape, size and functionality and collection containers included tubes, cassettes and cardboard sleeves of varying shapes and sizes. (For further information and photographs of the test kits presented in consultations, see Supplementary File 1, available from: figshare.com/articles/online_resource/Supplementary_File_1_Goodwin_et_al_2022/19388600).
Procedure
Each participant took part in a private consultation with a registered nurse (BV) at a time convenient to them. During the consultation, participants were presented with the nine FOBT kits and given the opportunity to inspect the contents, instructions and labelling. Because of social distancing requirements during the COVID-19 pandemic, nine of the 25 consultations were conducted by video link rather than face to face. During the video consultations, BV presented high-quality images of each of the nine FOBT kits and their contents, showed the packaging and contents of each kit including paperwork and stool collection implements, and guided the participant though an inspection of each kit’s features and the instructions.
Using a semi-structured interview script (see Supplementary File 2, available from: figshare.com/articles/online_resource/Supplementary_File_2_Goodwin_et_al_2022/19388627), participants were asked to consider the features of each kit (including packaging, collection tools, collection vessels, instructions, etc.) and the degree to which each feature might act as a barrier to kit use and/or return. Participants were also asked about their history of receipt and use of the NBCSP kit, any other barriers to use and any suggestions for improvements to the FOBT kit currently used in the national program.
Interviews ranged from 30 minutes to 1 hour. With permission, interviews were audio-recorded, transcribed and de-identified. Detailed notes were taken during each session for use when full audio was not captured on recording.
Coding and analysis
Interviews were coded sequentially; from interview number 18 onwards, no novel responses were identified during the coding, suggesting data saturation had been reached.16
Content analysis was used to describe and interpret participants’ opinions and perspectives.17 A conventional approach was chosen as it allows for raw qualitative data to be summarised into categories or themes based on reasonable interpretations.18 This method also allows for a focus on subject and context, highlighting similarities and differences in participants’ experiences.19 All participant responses that mentioned a specific feature that could act as a barrier or a facilitator to using the kit were allocated a code. These were often expressed as preferences or dislikes regarding features of certain kits. Resulting codes were categorised according to the element of the kit they related to (e.g., packaging, collection device, kit return protocol). Coding of the first four transcripts was conducted by BG. Through discussion with BV, the coding and structure of codes were refined, and BV coded the remaining consultations using this structure with the option of adding new codes as they emerged. BG reviewed and refined coding by collapsing some similar codes with largely identical sets of responses. To check intercoder reliability, LM reviewed the coding structure and categorisations of response data and agreed with all decisions made. Responses relating to each element of the kit were then summarised.
Results
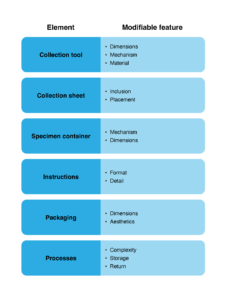
Six elements of the home bowel cancer screening kits were identified, each having two or more modifiable features that may facilitate or act as barriers to the use of home bowel cancer screening kits (see Figure 1). Three elements – the collection tool, the collection sheet and the specimen container – pertained to the equipment provided for stool collection and storage. The remaining three were the instructions, the packaging of the kit, and the processes or the steps involved in collecting, storing and returning the samples. A table of exemplar quotes pertaining to each element is presented in Supplementary File 3 (available from figshare.com/articles/online_resource/Supplementary_File_3_Goodwin_et_al_2022/19388642).
Figure 1. Elements and modifiable features of home bowel cancer screening kits that may facilitate or act as barriers to use (click to enlarge)
Collection equipment
Collection tool
Most participants stated that the handle of the collection tool should be longer. Basic wooden spatulas, referred to by some as “paddle pop sticks”, were often preferred over the tools provided in the current NBCSP collection kit due to their extra length and width and some were pleased that they were made of biodegradable material. However, many expressed that the process of smearing the stool to collect a sample would be challenging and some showed concern that the spatulas might break. Many commented that they liked the grip on the handles of the tools previously distributed in the NBCSP kit, also preferring their slightly longer handles. Several participants mentioned that the collection tools with a “grooved tip” mechanism helped to collect a sample, with some suggesting the grooves should be wider and deeper to increase the ease of collection, ensuring a big enough sample. Some participants felt the brush mechanism for collecting the sample in some kits was preferable to “scooping up” particles of the stool, and many commented that they thought the length of the handle of the brush tool was ideal. However, most participants tended not to like the idea of depositing a sample onto a cardboard collection pad using a brush, feeling it was a less hygienic and less reliable method.
Collection sheet
Almost all participants agreed that a collection sheet was an important inclusion in a home screening kit, with several participants checking that they were biodegradable and mentioning this was a necessity. Concerns about kits that did not have collection sheets included the potential contamination of the sample and physical challenges collecting the sample. Many felt that without the collection sheet it would be difficult to target the stool and one may end up “chasing it around the bowl”. Opinions varied on the optimal design of the collection sheet in terms of where it should be placed. Most participants preferred the type of collection sheet that is placed inside the bottom of the toilet bowl above the water, such as the sheet provided in the current NBCSP kit, but often noted the handle of the collection tool needed to be longer to accommodate for reaching into the toilet with this collection sheet design. Other participants favoured attaching the collection sheet to the seat or the top of the toilet bowl. Many suggested that this design was optimal when the sheet dipped into the bowl, rather than sitting straight across the top of the toilet. Also, most agreed that although collection sheets that attached to the toilet seat were useful, the ones available were too narrow and users risked “missing” the sheet when depositing their stool.
Specimen container
Cardboard specimen pads were generally not preferred or were not viewed as ideal as participants saw them as unhygienic and insecure. Participants mentioned concerns about accidentally touching the stool sample when closing the cardboard flap and that the flap may not remain closed, exposing the stool sample while waiting for second and third samples to be deposited.
The test tubes used in the current NBCSP were preferred by many over other collection tubes due to them being separate secure containers with clip-on (as opposed to twist-on) lids. However, several participants expressed hygiene concerns over the fact that the handle on the current NBCSP tool doubled as the lid for the specimen container, preferring to drop the whole collection device into the tube and screw on a separate lid. Several participants liked the idea of colour coding lids to distinguish between the first and second sample.
It was commonly suggested that the specimen tube included in the current NBCSP kit was too small, with reference to the opening. Many were concerned that inserting the sample into the tube took a lot of precision and risk of “missing” the insertion point was high. Many also commented on the difficulty involved in writing details on the sticker label while it was stuck on the tube and suggested it would be easier to write the details on a (bigger) sticker and attach it to the tube themselves, or for containers to arrive with “prefilled” stick-on labels or barcodes to attach.
Several participants commented that having several layers covering the container itself made them feel more comfortable about storing it in the refrigerator and preferred kits that provided “numerous barriers for protection” from the sample. The earlier version of the NBCSP kit (i.e., kit number 8 in Supplementary File 1, available from: figshare.com/articles/online_resource/Supplementary_File_1_Goodwin_et_al_2022/19388600) was distributed to recipients of the program for several years, before it was replaced by the current kit in around 2018. This previous kit was often referred to as the optimal design in terms of protection, with a collection tube with a screw top into which the entire collection tool is inserted, plus the ziplock bag.
Instructions
There was a clear consensus among participants that concise, step-by-step instructions with large font and descriptive, clearly numbered images were preferred over text-heavy, overly detailed instructions. Several participants implied that they would most likely not read through text, relying on pictures to guide them and some suggested that images were more suitable than text for people for whom English is a second language. Participants favoured instruction pamphlets with large, coloured images that depicted the environment (i.e., bathroom, toilet and person) as opposed to just the test equipment itself.
Some kits, including the current NBCSP, contained detailed information booklets about bowel cancer, screening and risk. Responses regarding the inclusion of extra information about bowel cancer such as signs and symptoms, risk and details of pathology processes were mixed. Some people were “put off” by the extra text included in some kits, preferring to be presented only with the instructions they required to complete the screening test. Whereas others appreciated being provided with additional information for increasing their knowledge and providing context for bowel cancer screening. Even those who stated that they would be interested in reading extra information about bowel cancer suggested it be brief, positively framed, and include references to other sources for further reading.
Packaging
Participants showed a preference for smaller kit packaging, tending to favour a compact, colourful box. It was noted that most of the kits in the study would not fit in letterboxes and envelopes were often preferred over boxes for that reason. This was largely due to concerns about the package being left where it might become damaged or seen by neighbours.
Participants also suggested that the size and design of the current NBCSP kit packaging elicited thoughts of medical procedures or hospital, making recipients feel “overwhelmed” and confronted by the potential seriousness of the contents, particularly when the word “cancer” or pictures of a colon were printed on the box.
Packaging with fewer logos and less text conveyed the perception that the test was “quick, easy and simple” and was less conspicuous for storage in shared areas. Whereas some participants preferred the aesthetics of the envelope currently distributed in the Australian program, as it conveyed a sense of professionalism and rigour; they also found the blue and white colouring “calmer” as opposed to some red packaging used in other kits that indicated alert or danger.
Processes
Some participants reported that the amount of equipment and number of steps to be taken in some kits made them daunting or overwhelming. For example, some commented on the complexity of the previous version on the NBCSP kit and “all the different levels of things you had to put in”. Participants tended to be undecided about whether they prefer test kits that required fewer samples. Although single sample tests were preferred over those requiring two or three samples due to ease and convenience, many participants conceded that if the reliability of the test was improved with more samples, they were willing to comply and provide two or even three samples.
Most participants viewed dietary restrictions prior to completing an FOBT kit as a barrier to completing a test with some suggesting it would deter them completely, particularly those who routinely consumed the restricted foods (e.g., red meat) or took certain medications. Others explained that restricting their diet would require a lot of planning ahead and would therefore delay the process of completing the test, but believed they would find a way to comply if it resulted in a more accurate test.
One common hygiene concern among participants was the method of disposal of equipment used in stool collection. Some participants were comfortable with the idea of wrapping collection tools, containers and in some cases used toilet paper in a plastic bag and disposing of them in an outdoor bin, whereas many strongly preferred to be able to “flush away” any parts of the kit that were no longer needed.
Many participants reported hygiene concerns about storing a stool sample in their refrigerator. Participants acknowledged, however, that it was likely a necessity in the hot Australian climate that samples be stored in the fridge and many were willing to do so given the sample was securely sealed. Nevertheless, others struggled, some admittedly irrationally, with the idea of their stool sample being stored in the vicinity of food (particularly food that other people had access to) regardless of how securely it was sealed.
Preferences regarding posting the kit back versus getting an immediate result (i.e., self-testing) were varied and based on factors such as where participants lived, the perceived accuracy of the results and their level of anxiety. Many liked the idea of receiving an immediate result to avoid delays and the anxiety of waiting for a result; a preference mostly expressed by those who had not participated in the NBCSP. Some participants preferred the self-testing method as it did not involve another person or because there was no need to store or post the sample and it was more perceived as discreet and private. Several participants expressed concern over the panic one may experience in receiving a positive result or the likelihood they may dismiss it without the support of a medical professional. For this reason, it was suggested that self-testing kits should have clear advice about what a positive result does and does not mean, and advice on next steps. Most felt more secure knowing their results were determined by professionals in a laboratory and many stated it was important that there was a record of their result with their GP, so that they could be followed up with reminders for further care. However, several male and female participants likened the testing cassettes provided with these self-test kits to pregnancy tests, indicating that they looked familiar and simple to use.
Finally, several people reported feeling uncomfortable about posting stool samples back through the mail service as they felt it was unhygienic or were concerned about maintaining the integrity of the sample. One rural participant reported concern with the practicality of posting their sample as they lived in a small community without a post office.
Discussion
The current study took a consumer-centred approach to identifying features of home bowel cancer testing kits that may facilitate and promote their use in population screening programs. The findings strongly suggest that processes, collection tools and storage devices that reduce the user’s proximity to, or potential contact with, faecal matter are key to facilitating sample collection and that various aesthetic features of packaging may also reduce cognitive and/or attitudinal barriers to program participation.
Our findings support changes to home bowel cancer screening kits that simplify processes, limit the chance of touching the stool and remove the double handling of collection equipment. This is in line with previous studies that have identified hygiene as a key concern for kit users.8,20,21 Evidence suggests that, where applicable, reducing the number of samples required from three to two, removing dietary restrictions prior to sampling, and including gloves with the kit can result in increased participation.9 Some facilitating features identified by consumers in the current study may be less effective than others. For example, although the inclusion of a collection sheet was preferred by almost all participants in the current study, previous trials have demonstrated that its inclusion in unlikely to increase participation in population bowel cancer screening programs.22,23
In terms of packaging and associated materials, small, minimalistic packaging, with positively framed messaging and clear step-by-step instructions were preferred. Trials assessing the effectiveness of simplifying home bowel cancer screening instructions have yielded mixed results9, suggesting this method may not be effective in all contexts. This may also be reflected in the mixed opinions in the current study regarding the inclusion of extra information about bowel cancer. One solution to this may be to provide the option of extra information via referrals or weblinks within the kit, rather than inserting detailed booklets. Further research may be needed to identify the optimal amount and presentation of information accompanying home test kits.
Findings here suggest that the home screening kit currently distributed through the NBCSP features many elements of an optimal kit. That is, the aesthetics, packaging, testing and posting processes and the clear instructions provided were endorsed by many participants. However, various modifications to the stool collection tools may help to alleviate hygiene concerns and improve overall usability. These include lengthening the collection tool by as much as three times the current length; providing a wider opening to the test tube and a separate lid; and providing a storage system for the completed sample that offers more protection for the handler from making contact with the stool sample. Ultimately, a storage mechanism that does not require the user to re-open the bag that the samples are stored in and/or one that removes the need for refrigeration would be ideal.
Strengths and limitations
This study was the first to our knowledge to consult directly with consumers regarding their preferences on various government-issued and commercially available home bowel cancer screening kits. There is potential selection bias inherent in a volunteer sample, however, the sample comprised a reasonably even distribution of gender, age group and level of experience with kit use. While the recommendations that stem from this study are largely applicable to the current NBCSP kit distributed in Australia, they also inform international programs and home test kit design more broadly. For example, three sample stool tests and cardboard specimen pads are still widely available for use in Australia and international settings. Willingness to engage in testing and screening behaviour could be increased through replacing these methods with more user-friendly alternatives.
It is important to apply caution in generalising these findings to the wider population given the small sample size and the fact that the sample exclusively represents computer users who may have different views and preferences on the use of technology and devices when compared with those who don’t use computers. Finally, it is acknowledged that these findings reflect individual opinions, perceptions and intentions, and evidence of the effectiveness of the suggested kit modifications in increasing participation above levels achieved with current kits will require randomised control trials.
Conclusion
Physical barriers to the use of home bowel cancer screening kits are often overlooked, although there is clear evidence that such barriers play a key role in deterring participation in population bowel cancer screening programs.8 Findings from this study provide several consumer-driven recommendations for improving the acceptability and useability of home sample collection kits for distribution in population bowel cancer screening programs in both Australia and international settings. Relatively minor modifications including changing the dimensions and aesthetics of collection tools and kit packaging may result in an increased willingness to participate in bowel cancer screening.
Peer review and provenance
Externally peer reviewed, not commissioned.
Copyright:
© 2022 Goodwin et al. This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International Licence, which allows others to redistribute, adapt and share this work non-commercially provided they attribute the work and any adapted version of it is distributed under the same Creative Commons licence terms.
References
- 1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. CrossRef | PubMed
- 2. Australian Institute of Health and Welfare. Cancer data in Australia. Canberra; AIHW: 2020 [cited 2022 Mar 15]. Available from: www.aihw.gov.au/reports/cancer/cancer-data-in-australia/contents/summary
- 3. Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, Meester RG, Barzi A, et al. Colorectal cancer statistics, 2017. CA Cancer J Clin. 2017;67(3):177–93. CrossRef | PubMed
- 4. Lew J-B, St. John DJB, Macrae FA, Emery JD, Ee HC, Jenkins MA, et al. Evaluation of the benefits, harms and cost-effectiveness of potential alternatives to iFOBT testing for colorectal cancer screening in Australia. Int J Cancer. 2018;143(2):269–82. CrossRef | PubMed
- 5. Navarro M, Nicolas A, Ferrandez A, Lanas A. Colorectal cancer population screening programs worldwide in 2016: an update. World J Gastroenterol. 2017;23(20):3632–42. CrossRef | PubMed
- 6. Australian Institute of Health and Welfare. National Bowel Cancer Screening Program monitoring report 2021. Canberra: AIHW; 2021 [cited 2021 Jul 22]. Available from: www.aihw.gov.au/reports/cancer-screening/nbcsp-monitoring-report-2021/summary
- 7. Lew J-B, St John DJB, Xu X-M, Greuter MJ, Caruana M, Cenin DR, et al. Long-term evaluation of benefits, harms, and cost-effectiveness of the National Bowel Cancer Screening Program in Australia: a modelling study. Lancet Public Health. 2017;2(7):e331–40. CrossRef | PubMed
- 8. Goodwin BC, Myers L, Ireland MJ, March S, Ralph N, Dunn J, et al. Barriers to home bowel cancer screening. Psychooncology. 2021;30(10):1756–64. CrossRef | PubMed
- 9. Goodwin BC, Ireland MJ, March S, Myers L, Crawford-Williams F, Chambers SK, et al. Strategies for increasing participation in mail-out colorectal cancer screening programs: a systematic review and meta-analysis. Syst Rev. 2019;(8):257. CrossRef | PubMed
- 10. Myers L, Goodwin B, Ralph N, Castro O, March S. Implementation strategies for interventions aiming to increase participation in mail-out bowel cancer screening programs: a realist review. Front Oncol. 2020;10:543732. CrossRef | PubMed
- 11. Flach JM, Dominguez CO. Use-centered design: integrating the user, instrument, and goal. Ergon Des. 1995;3(3):19–24. CrossRef
- 12. Peck RB, Lim JM, van Rooyen H, Mukoma W, Chepuka L, Bansil P, et al. What should the ideal HIV self-test look like? A usability study of test prototypes in unsupervised HIV self-testing in Kenya, Malawi, and South Africa. AIDS Behav. 2014;18(4):422–32. CrossRef | PubMed
- 13. Surabattula D, Harvey CM, Aghazadeh F, Rood J, Darisipudi A. Usability of home cholesterol test kits and their impact on patients’ decision. International Journal of Industrial Ergonomics. 2009:39(1):167–73. CrossRef
- 14. Feufel MA, Schneider TR, Berkel HJ. A field test of the effects of instruction design on colorectal cancer self-screening accuracy. Health Educ Res. 2010;25(5):709–23. CrossRef | PubMed
- 15. Myers L, Goodwin B, Ralph N, Castro O, March S. Implementation strategies for interventions aiming to increase participation in mail-out bowel cancer screening programmes: a realist review. Front Oncol. 2020;10:1799. CrossRef | PubMed
- 16. Glaser BG, Strauss AL. The:discovery of grounded theory: strategies for qualitative research. New York: Routledge; 2017.
- 17. Elo S, Kääriäinen M, Kanste O, Pölkki T, Utriainen K, Kyngäs H. Qualitative content analysis: a focus on trustworthiness. SAGE Open. 2014;4(1):2158244014522633. CrossRef
- 18. Zhang Y, BM Wildemuth. Qualitative analysis of content.Human Brain Mapping. 2005;30(7). Article
- 19. Graneheim UH, Lindgren B-M, Lundman B. Methodological challenges in qualitative content analysis: a discussion paper. Nurse Educ Today. 2017;56:29–34. CrossRef | PubMed
- 20. Goodwin BC, March S, Crawford-Williams F, Chambers SK, Dunn J. “I’m not doing that.” An in-depth examination of nonparticipation in mail-out bowel cancer screening programs. Transl Behav Med. 2020:10(6)1515–24. CrossRef | PubMed
- 21. Gordon NP, Green BB. Factors associated with use and non-use of the Fecal Immunochemical Test (FIT) kit for colorectal cancer screening in response to a 2012 outreach screening program: a survey study. BMC Public Health. 2015;15:546. CrossRef | PubMed
- 22. Denters MJ, Deutekom M, Bossuyt PM, Fockens P, Dekker E. A feces collection paper does not enhance participation in a fecal immunochemical test-based colorectal cancer screening program: randomized clinical trial. Eur J Cancer Prev. 2013;22(4):299–304. CrossRef | PubMed
- 23. Morling JR, Barke AN, Chapman CJ, Logan RF. Could stool collection devices help increase uptake in bowel cancer screening programmes? J Med Screen. 2018(4):174–7. CrossRef | PubMed