Abstract
Objectives: Electronic cigarettes (ECs) have recently become popular around the world, and their safety is being widely discussed in the scientific literature. Previous studies have examined the chemicals in e-liquids and vapour, and demonstrated that the aerosol from ECs can contain toxic chemicals that are harmful to health. However, little is known about the potential adverse health effects of passive exposure to EC vapour. The aim of this paper is to summarise and review all studies that have examined potential adverse health effects of passive exposure from inhaling EC vapour. Specifically, our research objectives were to describe 1) the absolute impact of passive exposure from inhaling vapour when compared with background, and 2) the relative impact of passive exposure from inhaling vapour when compared with passive exposure from inhaling conventional cigarette smoke.
Methods: A systematic review was conducted to identify articles published from 1996 to 10 September 2015 from Embase, Ovid MEDLINE and PreMEDLINE. Papers eligible for inclusion had to be written in English, study health effects from passive exposure to EC vapour in animals or humans, test or analyse the EC vapour directly or in the ambient air (with an inference made about passive or second-hand vapour exposure). The review was conducted using the PRISMA guidelines for reporting on systematic reviews. We identified 312 studies, and 16 were relevant for inclusion in our review.
Results: A variety of study designs were used to investigate potential health risks from passive exposure to EC vapour. These included direct exposure studies involving humans and animals, and indirect exposure studies using volunteer EC users or smoking machines. The majority of studies determined that passive exposure to EC vapour may pose a health risk to bystanders. All papers encountered a number of limitations.
Conclusion: Our review found that the absolute impact from passive exposure to EC vapour has the potential to lead to adverse health effects. The risk from being passively exposed to EC vapour is likely to be less than the risk from passive exposure to conventional cigarette smoke.
Full text
Introduction
Electronic cigarettes (ECs), also called e-cigarettes, e-cigs or electronic nicotine delivery systems (ENDS) are battery-powered devices that vaporise a liquid (also called e-liquid) into an aerosol. ECs come in a variety of designs and can be disposable or reusable. They typically consist of a battery, an airflow sensor to activate flow of power to the device, an aerosol generator and a solution (or e-liquid) storage area.1 Unlike conventional cigarette (CC) users who inhale smoke produced by burning tobacco, the EC user inhales an aerosol, which typically contains nicotine, propylene glycol and other chemicals.2 Inhaling the aerosolised e-liquid is referred to as vaping.
In recent years, EC use has become more popular around the world. A survey in the US in 2014 investigating EC and tobacco use showed it was the most common product used by middle- and high-school students.3 The survey also showed that, among middle- and high–school students, EC use tripled from 2013 to 2014 (from 3.9% to 13.4% of students). In New South Wales (NSW), a survey showed that the prevalence of current EC users in 2014 was 1.3%, with 8.4% of people having tried an EC.4 The authors estimated that about 78 000 people were current EC users in NSW; this is relatively low compared with some other countries, including the US and the UK. It is unclear whether similar increases as observed in the US are to be expected in Australia.
Over the past few years, the public health literature has discussed use of ECs as a smoking reduction and cessation device, the possibility for ECs to undermine long-term efforts to denormalise smoking, and the safety and potential adverse health effects of ECs for users and bystanders.5,6 Recent reviews have found little or no evidence to support the use of ECs as a smoking reduction and cessation device6,7, and public health experts warn of the potential for ECs to normalise smoking as their use becomes more frequent.8
The most important safety concerns relating to ECs include exposure to nicotine, particulate matter (PM) and other chemical substances, and the safety of the device itself.9 Several studies have previously examined chemicals in e-liquids and vapour, and demonstrated that the aerosol from ECs can contain toxic chemicals that are harmful to health; the vapour is not merely ‘water vapour’ as has been claimed in the past.2,10,11 In one study, levels of chemicals in EC vapour were found to be 9–450 times lower than levels in CC smoke.10
Passive exposure to CC smoke, also called second-hand smoke or environmental tobacco smoke, has been extensively researched, and is well known to be hazardous to health.12 Passive exposure to EC vapour is not well studied because ECs are relatively new. Unlike CCs, ECs produce no secondary or side-stream emissions; therefore, passive exposure consists only of what the EC user exhales. Nevertheless, passive exposure to ECs remains a concern because of its potential adverse health effects for people who are involuntarily exposed.
This systematic review aims to describe and summarise all studies to date that have examined potential adverse health effects of passive exposure from inhaling EC vapour. Specifically, our research objectives were to separate results describing the absolute impact of passive exposure from inhaling EC vapour when compared with background (ambient air) and the relative impact of passive exposure from inhaling EC vapour when compared with passive exposure from inhaling CC smoke. Adverse health effects for EC users from directly inhaling the vapour were not considered in this review; however, passive exposure to vapour also affects EC users.
Methods
The review was conducted using the PRISMA guidelines for reporting on systematic reviews, where applicable.13 We searched Embase, Ovid MEDLINE and PreMEDLINE from 1996 to 10 September 2015 inclusive, using the following search terms: ‘electronic cigarette/s’ or ‘e-cigarette/s’ or ‘e cigarette/s’ or ‘electronic nicotine delivery’ or ‘vaping, vape or vaper/s’, combined with the search terms ‘passive’ or ‘secondhand’ or ‘second hand’ or ‘exposure’ or ‘exposed’ or ‘vulnerable’ or ‘nonuser/s’ or ‘non-user/s’.
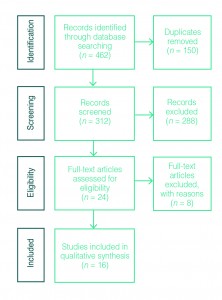
After limiting the search to English, we identified 462 studies. Duplicates accounted for 150 studies. We screened a total of 312 titles for relevance to ECs and health effects, excluding titles describing regulation, perceptions, advertisements and uptake studies (n = 137). The remaining 175 abstracts were reviewed.
To be considered for inclusion, the study had to:
- Look at health effects from passive exposure to EC vapour (animal or human), or
- Test or analyse the EC vapour directly, or test or analyse ambient air with EC vapour and make an inference with regard to passive (second-hand) vapour exposure.
Studies were also included if it was not possible to make a decision from the abstract on inclusion/exclusion criteria.
Studies were excluded if they looked at:
- Only health effects from direct exposure to the user
- In utero exposure from an EC vaping mother
- Third-hand exposure to EC vapour (i.e. from contaminated surfaces)
- Ingestion or dermal exposure to EC liquid.
Studies were also excluded if they did not include original data.
Agreement of the first two authors was necessary to exclude a paper.
This process yielded 24 abstracts for full-text review. Eight papers were excluded after the full text was read thoroughly. Reference lists of the full-text articles were examined to identify additional publications, but none were found. Figure 1 shows the paper selection process.
Figure 1. Paper selection process for systematic review, adapted from Moher et al. 200913 (click to enlarge)
Papers were reviewed and information was recorded on study design, participants, main results and conclusions, declared conflicts of interest, and study limitations.
Results
We found 16 relevant studies (including one conference abstract) that investigated potential adverse health effects from passive exposure to ECs (Table 1). For each study, we specified whether ECs were compared with background levels, CCs or both. The studies were grouped into four study designs:
- Direct passive exposure studies with human volunteers (n = 4)
- Direct passive exposure studies in animal models (n = 1)
- Indirect exposure studies with human volunteers (± smoking machine) using ECs (n = 7)
- Indirect exposure studies with no human volunteers (n = 4).
Table 1. Studies identified through systematic review
| Study type | Author and publication year | Study design | Results | Conflict of interest | Limitations |
| Direct passive exposure studies with human volunteers | Ballbè et al. (2014) | Observational exposure study | Nicotine: CC homes > EC homes > background Cotinine: raised with passive EC exposure; same as CC |
No | Small sample size Questionnaire: potential bias |
| Flouris et al. (2013) | Experimental repeated measure exposure study | Cotinine: raised with passive EC exposure; same as CC Lung function: decreased only after passive CC exposure |
No | Small sample size Smoking machine Short exposure |
|
| Flouris et al. (2012) | Experimental randomised crossover exposure study | Full blood count measures: increased only after passive CC exposure | No | Small sample size Smoking machine Short exposure |
|
| Tzatzarakis et al. (2013) | Experimental repeated measure exposure study | Inflammatory markers: increased only after passive CC exposure | * | Limited information: abstract only Small sample size Short exposure |
|
| Direct passive exposure studies in animal models | McGrath-Morrow et al. (2015) | Animal exposure study | Mice exposed to EC: weighed less Mice exposed to EC with nicotine: elevated cotinine, impaired lung growth |
No | Applicability to humans: unclear Smoking machine |
| Indirect exposure studies with human volunteers (±smoking machine) using ECs | Czogala et al. (2013) | Indirect exposure study (using smoking machine) | Nicotine: elevated after EC use, but higher after CC use PM2.5: elevated after EC use, but higher after CC use |
Funding by EC manufacturer | Indirect study only Limited chemicals measured Input air not filtered, air exchange rates not realistic |
| Long (2014) | Indirect exposure study | Phenolics, carbonyls: only increased after CC use | Employee of tobacco company | Indirect study only Limited chemicals measured No actual concentrations |
|
| O’Connell et al. (2015) | Indirect exposure study | Propylene glycol, VOCs, formaldehyde, acetaldehyde: increased after EC use | Employees of tobacco company | Indirect study only | |
| Ruprecht et al. (2014) | Indirect exposure study | PM2.5, UFPs: highest after CC use; EC without nicotine higher than EC with nicotine | * | Indirect study only Only difference in concentrations |
|
| Saffari et al. (2014) | Indirect exposure study | B, K, La, Zn, Ni, Ag: increased after EC use | * | Indirect study only Comparison with outdoor air | |
| Schober et al. (2013) | Indirect exposure study | PM: increased after EC use, highest after e-liquids without nicotine 1,2-propanediol, glycerine, nicotine, PAHs, aluminium: increased after EC use |
No | Indirect study only | |
| Schripp et al. (2013) | Indirect exposure study | 1,2-propanediol: detected after EC use, higher after CC use UFP and PM2.5 in EC aerosol | No | Indirect study only | |
| Indirect exposure studies with no human volunteers | Colard et al. (2015) | Development and testing of air quality model | Model was good predictor Nicotine not a health concern |
Employees of tobacco company | Indirect exposure study using model |
| Geiss et al. (2015) | Indirect exposure study | Propylene glycol, glycerol, nicotine, carbonyls, aerosol particulates: detected after EC use | No | Indirect exposure study Smoking machine |
|
| McAuley et al. (2012) | Indirect exposure study | PM, nicotine, carbonyls, TSNAs, BTEX: lower after EC versus CC use | Member of National Vapers Club (funded study) | Indirect exposure study Smoking machine Cross-contamination |
|
| Pellegrino et al. (2012) | Indirect exposure study | PM: increased after EC use, higher after CC use | * | Indirect exposure study Smoking machine |
Ag = silver; B = boron; BTEX = benzene, toluene, ethylbenzene and xylenes; CC = conventional cigarette; EC = electronic cigarette; K = potassium; La = lanthanum; Ni = nickel; PAH = polycyclic aromatic hydrocarbon; PM = particulate matter; PM2.5 = fine particles, with a diameter smaller than 2.5 micrometres; TSNA = tobacco-specific nitrosamine; UFP = ultrafine particles, with a diameter smaller than 1 micrometre; VOC = volatile organic compound; Zn = zinc
* Conflict of interest was not specifically addressed in the paper.
Direct passive exposure studies with human volunteers
Four studies aimed to directly assess passive exposure in human volunteers.14-17 All four had a small number of volunteers. They compared volunteers passively exposed to EC vapour with non-exposed volunteers, and also separately compared volunteers passively exposed to EC vapour with volunteers passively exposed to CC smoke.
Ballbè et al.14 conducted an observational study including 54 nonsmoking volunteers from different homes (EC users, living with CC smokers, or nonsmoking home). The living-room air was sampled for nicotine for 7 days, and saliva and urine samples were collected from the volunteers after this week of exposure. The results showed significantly higher levels of airborne nicotine in homes with EC users than in nonsmoking control homes. In homes with CC smokers, airborne nicotine was significantly higher than in homes with EC users. Salivary and urinary cotinine levels were significantly lower in volunteers from nonsmoking control homes than in volunteers exposed to either EC vapour or CC smoke, with both the latter having elevated levels of cotinine. This showed that nonsmokers passively exposed to EC vapour absorb a similar amount of nicotine as nonsmokers passively exposed to CC smoke, despite the differing airborne nicotine concentrations.
In another experiment, Flouris et al.15 exposed 15 nonsmokers for 1 hour to second-hand CC smoke or EC vapour generated by a smoking machine. Serum cotinine and lung function measures were taken for each participant. No difference was found in lung function for the nonsmokers passively exposed to EC vapour compared with no exposure, but participants’ serum cotinine levels were raised, similar to volunteers passively exposed to CC smoke. The authors also published an earlier paper using the same experimental design but measuring complete blood count indices in volunteers.16 This study found that short-term passive EC exposure did not seem to lead to the inflammatory response that is seen in volunteers passively exposed to CC smoke – blood count measures were unchanged with EC exposure. The low-grade inflammatory response from exposure to CC is thought to be a step in the pathway to cardiovascular disease. These two studies demonstrate that participants exposed to EC vapour show the elevated serum cotinine levels that are seen in participants passively exposed to CC smoke; however, the short-term exposures to EC vapour did not elicit a reduction in lung function or an increase in inflammatory markers.
Tzatzarakis et al.17 studied a different set of inflammatory markers from EC exposure, including interleukins, vascular endothelial growth factor, tumour necrosis factor alpha, monocyte chemotactic protein-1 and epidermal growth factor. Little information was available on the authors’ study design because this publication was a conference abstract. The study involved 10 nonsmokers passively exposed to EC vapour for 1 hour, followed by measurement of inflammatory markers. As found by Flouris et al.16, this short-term passive exposure to EC vapour did not significantly affect inflammatory markers in the exposed subjects.
Direct passive exposure studies in animal models
Only one animal study was identified that specifically looked at passive exposure from ECs. McGrath-Morrow et al.18 studied the effect of passive EC exposure on newborn mice in the first 10 days of life. Animals were exposed to either room air (controls) or EC vapour with or without nicotine once or twice a day for 20 minutes. After 10 days, measurements showed that mice exposed to EC vapour (with or without nicotine) weighed significantly less than mice exposed to room air only. Mice exposed to vapour containing nicotine also showed impaired lung growth, and elevated plasma and urine cotinine levels.
Indirect passive exposure studies with human volunteers (± smoking machine) using ECs
Seven studies were identified that indirectly studied passive exposure by measuring chemical and toxicological compounds in the vapour produced by human volunteers using ECs.19-25 Three of the seven studies reported a conflict of interest (Table 1).19-21
Czogala et al.19 measured ambient levels of nicotine, fine particulate matter (PM2.5), carbon monoxide (CO) and selected volatile organic compounds (VOCs) in a ventilated exposure chamber whose contents were generated by a smoking machine or exhaled by volunteers who had either smoked CCs or used ECs. ECs were compared with background and with CCs. The results showed significantly elevated levels of nicotine and PM2.5 in the ambient air compared with background; however, levels were much lower than when generated from CCs. Interestingly, PM2.5 levels were higher after EC use by volunteers than when generated by the smoking machine (no difference was found for nicotine). The authors did not find significantly elevated levels of CO or VOCs from the use of ECs.
Long20 examined directly exhaled EC aerosols from volunteers captured on a glass fibre filter pad. Water and glycerine were the major components, with no significant amounts of carbonyl or phenolic compounds. Only small amounts of nicotine were detected (0.05% of the overall composition of the exhaled aerosol), which the authors stated were of no concern for bystanders.
O’Connell et al.21 measured a wide range of chemical elements and compounds in the ambient air of a room with three active EC users and two nonsmokers. The authors found that only some chemicals had detectable levels, and all were within indoor air quality guidelines. The authors concluded that there was no apparent risk to bystanders.
A study by Ruprecht et al.22 specifically investigated PM emissions from ECs compared with CCs and background levels. The authors documented a very small increase for PM2.5 and ultrafine particles (UFP) for an EC containing nicotine compared with background levels. However, when an EC was used without nicotine solution, the levels of PM2.5 and UFP were significantly higher than background. Overall, the authors found lower PM levels from ECs than from CCs. Nevertheless, they concluded that nicotine-free solutions may still pose a risk for bystanders.
A comprehensive study by Saffari et al.23 examined ambient air for a wide selection of particulate metals and organic compounds. When compared with background levels (outdoor air), this study found EC use did not increase levels of total PM, black carbon or polycyclic aromatic hydrocarbons (PAHs). When compared with CCs, the authors describe a decrease in total PM and PAHs in vapour generated by ECs. Nevertheless, the findings include the detection of a range of chemical elements after EC use – some potentially originating from the actual device rather than the e-liquid. Nickel, chromium and silver were found to be increased after EC use compared with CC use. The authors concluded that ECs were an improvement over CCs from a public health perspective; however, some ECs could contain toxic metals that may lead to second-hand exposure from EC consumption.
Schober et al.24 also conducted a comprehensive analysis of ambient air in a cafe-like setting with three volunteer EC users present simultaneously. Compared with background levels, EC use was found to significantly increase PM2.5, 1,2-propanediol, glycerine and nicotine. Potentially carcinogenic PAHs increased by 20% and aluminium by 2.4-fold. No comparisons were made with CC use. The paper concluded that ECs are not emission-free and impair indoor air quality, and that this is potentially a health concern.
Another study, by Schripp et al.25, examined PM2.5, UFP and formaldehyde emissions from ECs compared with background and CCs. The authors confirmed that ECs are a new source of VOCs, PM2.5 and UFP, and could be of concern for people passively exposed.
Indirect passive exposure study with no human volunteers
Four studies were identified that used either smoking machines or mathematical modelling to simulate EC use and research exposure from ECs.26-29 Two reported a conflict of interest (Table 1).26,28
Colard et al.26 developed an air quality model to predict bystander exposure to chemical constituents from EC vapour exhaled by EC users within an indoor environment. The model was tested with inputs from the study by Czogala et al.19 and found to predict findings accurately. The model was then used to predict nicotine exposure for a bystander in a small, shared office space, where one office worker is an EC user. The model predicts that a bystander would inhale 4–8 micrograms of nicotine per day, which the authors stated does not cause health concerns.
A study by Geiss et al.27 analysed EC vapour produced by a smoking machine using different nicotine concentrations and compared ECs with background; no comparison was made with CCs. Analysis from a glass fibre filter pad and gas sampling bag, and the air in the study chamber determined levels of propylene glycol, glycerol, nicotine, carbonyls and aerosol particulates in the vapour generated by ECs. The authors stated that carbonyl contribution from vaping is likely to be negligible. However, people may still be passively exposed to components of EC vapour, depending on the setting and number of ECs in use.
McAuley et al.28 conducted a comprehensive study using a smoking machine to test PM, nicotine, tobacco-specific nitrosamines (TSNAs), PAHs, glycols, VOCs and carbonyls emitted from ECs compared with CCs. EC vapour was found to contain either lower levels of the chemicals tested than smoke from CCs, or levels below the detection limit. A toxicological assessment was then undertaken using the levels of chemicals detected in vapour emitted by ECs. It concluded that there is no significant risk of harm to human health from exposure to the levels of tested chemicals.
Another experimental study using a smoking machine, by Pellegrino et al.29, examined PM and the chemical composition of EC vapour, comparing ECs with background and CCs. EC use showed an increase in PM compared with background, with levels slightly exceeding World Health Organization air quality guidelines for short-term exposure. However, the authors stated that these guidelines are based on daily mean concentrations. ECs were found to emit significantly lower amounts of PM than CCs. Overall, the authors concluded ECs have advantages when used instead of CCs; however, they cause passive exposure to a number of chemicals, which requires further evaluation.
Discussion
We reviewed 16 studies, with varying designs, investigating potential adverse health effects of passive exposure to EC vapours. Studies examining the composition of EC vapour or some of its aspects found that ECs are not emission-free.19-25,27-29 The majority of studies concluded that passive exposure to EC vapour may pose a health risk to bystanders.14,15,18,19,22-25,27,29 Two studies did not comment on the passive exposure risk16,17 and four concluded that their investigation showed no risk to bystanders.20,21,26,28
It is noted that those studies undertaken by tobacco employees or funded by the National Vapers Club concluded no apparent risk from ECs to bystanders. Those who did not declare a conflict of interest were more likely to draw conclusions that were more precautionary and/or suggested a potential risk from passive exposure to ECs, highlighting potential biases in the current literature.
When examining the absolute impact from passive exposure to EC vapour – that is, comparing EC vapour with background levels – EC vapour contains elevated levels of nicotine19,20,24 PM19,22,24,25,29 glycerine20,24 propylene glycol21,24, formaldehyde and acetaldehyde21, PAHs and metals.24 These studies demonstrate that EC vapours can contain harmful chemicals and have an impact on indoor air quality.
When examining the relative impact from passive exposure to EC vapour – that is, comparing EC vapour with CC second-hand smoke – EC vapour contains much lower levels of most compounds measured.19,20,22,23,25,28,29 The exceptions are nickel and silver, which were higher in EC emissions than in CC smoke.23 This confirms that CCs pose a greater risk to the bystander than passive exposure to ECs.
Adverse health effects from exposure to nicotine and PM have been widely discussed in the literature.30-35 Epidemiological evidence from environmental studies has demonstrated adverse health effects from short-term and long-term exposures to PM, especially the smaller fraction of PM2.5, even at very low concentrations.32-34 Adverse health effects from exposure to PM2.5 include an increase in cardiovascular and respiratory diseases, as well as an increase in mortality from all causes.32,33 Nicotine has also been shown to have adverse health effects from short-term and long-term exposure.30,31 A recent review examined the effect of nicotine on the developing human, and concluded that nicotine exposure during vulnerable periods of brain and lung development, such as during pregnancy, childhood and adolescence, can have detrimental effects.35 Since EC vapour has been shown to contain PM as well as nicotine (when e-liquid with nicotine is used), passive exposure to EC vapour has the potential to cause adverse health effects. Chronic exposure, especially of infants and children in residential settings, would be of particular concern.
Adverse health effects have also been observed from exposure to some of the other chemicals that have been identified in e-liquids and vapour.10,36-38 For example, exposure to carbonyl compounds such as formaldehyde can cause irritation in acute settings and has been shown to cause nasopharyngeal cancers in humans with chronic exposure.36,38 Exposure to some heavy metals can cause organ toxicity37,38, and exposure to VOCs can cause irritation or cause cancer in long-term exposure settings.10,38 However, the levels of these chemicals are much lower in EC vapour than in CC smoke, and it is unclear whether these levels have adverse effects on passive bystanders.10,38 Nevertheless, little is known about what the effects may be with chronic passive exposure, and caution is warranted.38
All the studies examined have limitations. The studies that involved animal and human direct passive exposure only investigated short-term effects of exposure to EC vapour. Further, they had very small sample sizes. It is unclear how the animal study relates to humans; however, the findings of increased cotinine levels in neonate mice18 were replicated in two human passive exposure studies.14,15
All studies used only a maximum of three different brands of ECs, a limited number of e-liquids, and a limited number of measurement scenarios or repeat measurements. It is uncertain whether conclusions about EC safety for bystanders can be made on this basis when a wide variety of ECs and e-liquids are available on the market.5 It has also been shown that EC emissions can vary with differences in the battery power of the EC device39 and in the way people vape.40 It is questionable whether smoking machines are able to replicate human vaping behaviour41, and it is uncertain whether results from studies relying on this method are trustworthy. Further, there are no validated standard methods of testing EC vapour, and the reported concentrations of constituents may vary with the measurement techniques and sampling design used in studies.
Most studies measured emissions from one EC user (or smoking machine) only, with the exposure lasting from minutes to 1 hour. This type of scenario may be helpful for situations where people are exposed to the occasional EC user; however, such studies would not be useful to determine the risk to bystanders in other situations where many people are vaping simultaneously in enclosed spaces, such as in nightclubs, bars or cafes. In addition, risk patterns may change with cumulative exposure.
None of the studies looked at potential long-term impacts from exposure to EC vapour. Further, it is important to consider the impact that EC vapour may have on vulnerable population groups, such as children, pregnant women and people with chronic respiratory or cardiovascular conditions.
There is an urgent need to conduct further research to fill the knowledge gaps regarding passive exposure to EC vapour.
Conclusion
Although more research is required, current evidence regarding passive exposure to EC vapours shows the potential for health impacts. Those passively exposed to the vapours of EC users are exposed to numerous pollutants at levels above background and at concentrations that are associated with potential adverse health effects. The risk from being passively exposed to EC vapour is likely to be less than the risk from passive exposure to CC smoke.
Copyright:
© 2016 Hess et al. This article is licensed under the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International Licence, which allows others to redistribute, adapt and share this work non-commercially provided they attribute the work and any adapted version of it is distributed under the same Creative Commons licence terms.
References
- 1. Brown CJ, Cheng JM. Electronic cigarettes: product characterisation and design considerations. Tob Control. 2014;23:ii4–10. CrossRef | PubMed
- 2. Cheng T. Chemical evaluation of electronic cigarettes. Tob Control. 2014;23:ii11–17. CrossRef | PubMed
- 3. Arrazola RA, Singh T, Corey CG, Husten CG, Neff LJ, Apelberg BJ, et al. Tobacco use among middle and high school students – United States, 2011–2014. Morbidity and Mortality Weekly Report. 2015;64(14):381–5 [cited 2015 Mar 1]. Available from: www.cdc.gov/mmwr/preview/mmwrhtml/mm6414a3.htm
- 4. Harrold TC, Maag AK, Thackway S, Mitchell J, Taylor LK. Prevalence of e-cigarette users in New South Wales. Med J Aust. 2015;203(8):326. CrossRef | PubMed
- 5. Pisinger C, Dossing M. A systematic review of health effects of electronic cigarettes. Prev Med. 2014;69:248–60. CrossRef | PubMed
- 6. Grana R, Benowitz N, Glantz SA. E-cigarettes: a scientific review. Circulation. 2014;129(19):1972–86. CrossRef | PubMed
- 7. McRobbie H, Bullen C, Hartmann-Boyce J, Hajek P. Electronic cigarettes for smoking cessation and reduction. Cochrane Database System Rev. 2014;12:Cd010216. CrossRef | PubMed
- 8. Watson MC, Forshaw M. Why we shouldn’t normalise the use of e-cigarettes. BMJ. 2015;351:h3770. CrossRef | PubMed
- 9. NSW Health. Are electronic cigarettes and e-liquids safe? Sydney: NSW Government; 2014 [cited 2016 Feb 15]. Available from: www.health.nsw.gov.au/tobacco/Factsheets/e-cigs-are-they-safe.pdf
- 10. Goniewicz ML, Knysak J, Gawron M, Kosmider L, Sobczak A, Kurek J, et al. Levels of selected carcinogens and toxicants in vapour from electronic cigarettes. Tob Control. 2014;23(2):133–9. CrossRef | PubMed
- 11. Williams M, Villarreal A, Bozhilov K, Lin S, Talbot P. Metal and silicate particles including nanoparticles are present in electronic cigarette cartomizer fluid and aerosol. PLoS One. 2013;8(3):e57987. CrossRef | PubMed
- 12. Publications and Reports of the Surgeon General. The health consequences of involuntary exposure to tobacco smoke: a report of the Surgeon General. Atlanta, GA: US Department of Health and Human Services, Centers for Disease Control and Prevention, Coordinating Center for Health Promotion, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2006 [cited 2016 Mar 1]. Available from: www.surgeongeneral.gov/library/reports/secondhandsmoke/fullreport.pdf
- 13. Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009). Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. CrossRef | PubMed
- 14. Ballbè M, Martínez-Sánchez JM, Sureda X, Fu M, Pérez-Ortuño R, Pascual JA, et al. Cigarettes vs. e-cigarettes: passive exposure at home measured by means of airborne marker and biomarkers. Environ Res. 2014;135:76–80. CrossRef | PubMed
- 15. Flouris AD, Chorti MS, Poulianiti KP, Jamurtas AZ, Kostikas K, Tzatzarakis MN, et al. Acute impact of active and passive electronic cigarette smoking on serum cotinine and lung function. Inhal Toxicol. 2013;25(2):91–101. CrossRef | PubMed
- 16. Flouris AD, Poulianiti KP, Chorti MS, Jamurtas AZ, Kouretas D, Owolabi EO, et al. Acute effects of electronic and tobacco cigarette smoking on complete blood count. Food Chem Toxicol. 2012;50(10):3600–3. CrossRef | PubMed
- 17. Tzatzarakis MN, Tsitoglou KI, Chorti MS, Poulianiti KP, Jamurtas AZ, Koutedakis Y, et al. Acute and short term impact of active and passive tobacco and electronic cigarette smoking on inflammatory markers. Toxicol Lett. 2013;221 Suppl: S86. CrossRef
- 18. McGrath-Morrow SA, Hayashi M, Aherrera A, Lopez A, Malinina A, Collaco JM, et al. The effects of electronic cigarette emissions on systemic cotinine levels, weight and postnatal lung growth in neonatal mice. PLoS ONE. 2015;10(2):e0118344. CrossRef | PubMed
- 19. Czogala J, Goniewicz ML, Fidelus B, Zielinska-Danch W, Travers MJ, Sobczak A. Secondhand exposure to vapors from electronic cigarettes. Nicotine Tob Res. 2014;16(6):655–62. CrossRef | PubMed
- 20. Long GA. Comparison of select analytes in exhaled aerosol from e-cigarettes with exhaled smoke from a conventional cigarette and exhaled breaths. Int J Environ Res Public Health. 2014;11(11):11177–91. CrossRef | PubMed
- 21. O'Connell G, Colard S, Cahours X, Pritchard J. An assessment of indoor air quality before, during and after unrestricted use of e-cigarettes in a small room. Int J Environ Res Public Health. 2015;12(5):4889. CrossRef | PubMed
- 22. Ruprecht AA, De Marco C, Pozzi P, Munarini E, Mazza R, Angellotti G, et al. Comparison between particulate matter and ultrafine particle emission by electronic and normal cigarettes in real-life conditions. Tumori. 2014;100(1):e24–7. PubMed
- 23. Saffari A, Daher N, Ruprecht A, De Marco C, Pozzi P, Boffi R, et al. Particulate metals and organic compounds from electronic and tobacco-containing cigarettes: comparison of emission rates and secondhand exposure. Environ Sc Process Impacts. 2014;16(10):2259–67. CrossRef | PubMed
- 24. Schober W, Szendrei K, Matzen W, Osiander-Fuchs H, Heitmann D, Schettgen T, et al. Use of electronic cigarettes (e-cigarettes) impairs indoor air quality and increases FeNO levels of e-cigarette consumers. Int J Hyg Environ Health. 2014;217(6):628–37. CrossRef | PubMed
- 25 Schripp T, Markewitz D, Uhde E, Salthammer T. Does e-cigarette consumption cause passive vaping? Indoor Air. 2013;23(1):25–31. CrossRef | PubMed
- 26. Colard S, Connell G, Verron T, Cahours X, Pritchard J. Electronic cigarettes and indoor air quality: a simple approach to modeling potential bystander exposures to nicotine. Int J Env Res Public Health. 2014;12(1):282–99. CrossRef | PubMed
- 27. Geiss O, Bianchi I, Barahona F, Barrero-Moreno J. Characterisation of mainstream and passive vapours emitted by selected electronic cigarettes. Int J Hyg Environ Health. 2015;218(1):169–80. CrossRef | PubMed
- 28. McAuley TR, Hopke PK, Zhao J, Babaian S. Comparison of the effects of e-cigarette vapor and cigarette smoke on indoor air quality. Inhal Toxicol. 2012;24(12):850–7. CrossRef | PubMed
- 29. Pellegrino RM, Tinghino B, Mangiaracina G, Marani A, Vitali M, Protano C, et al. Electronic cigarettes: an evaluation of exposure to chemicals and fine particulate matter (PM). Ann Ig. 2012;24(4):279–88. PubMed
- 30. Centers for Disease Control and Prevention. How tobacco smoke causes disease: the biology and behavioral basis for smoking-attributable disease: a report of the Surgeon General. Atlanta, GA: Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2010 [cited 2016 Mar 1]. Available from: www.ncbi.nlm.nih.gov/books/NBK53017/
- 31. Office of the Surgeon General. The health consequences of smoking – 50 years of progress: a report of the Surgeon General. Rockville, MD: US Department of Health and Human Services, Public Health Service; 2014 [cited 2016 Mar 1]. Available from: www.surgeongeneral.gov/library/reports/50-years-of-progress/full-report.pdf
- 32. Pope CA 3rd, Dockery DW. Health effects of fine particulate air pollution: lines that connect. J Air Waste Manag Assoc. 2006;56(6):709–42. CrossRef | PubMed
- 33. US Environmental Protection Agency. Final Report: Integrated science assessment for particulate matter. Washington, DC: United States Environmental Protection Agency; 2009 [cited 2016 Mar 23]. Available from: cfpub.epa.gov/ncea/risk/recordisplay.cfm?deid=216546
- 34. HEI Review Panel on Ultrafine Particles. Understanding the health effects of ambient ultrafine particles. HEI Perspectives 3. Boston, MA: Health Effects Institute; 2013 [cited 2016 Mar 1]. Available from: pubs.healtheffects.org/getfile.php?u=893
- 35. England LJ, Bunnell RE, Pechacek TF, Tong VT, McAfee TA. Nicotine and the developing human: a neglected element in the electronic cigarette debate. Am J Prev Med. 2015;49(2):286–93. CrossRef | PubMed
- 36. International Agency for Research on Cancer. IARC monographs on the evaluation of carcinogenic risks to humans, volume 100F. Formaldehyde. Lyon, France: IARC; 2012 [cited 2016 18 Feb]. Available from: monographs.iarc.fr/ENG/Monographs/vol100F/mono100F-29.pdf
- 37. Järup L. Hazards of heavy metal contamination. Br Med Bull. 2003;68(1):167–82. CrossRef | PubMed
- 38. Bhatnagar A, Whitsel LP, Ribisl KM, Bullen C, Chaloupka F, Piano MR, et al. Electronic cigarettes: a policy statement from the American Heart Association. Circulation. 2014;130(16):1418–36. CrossRef | PubMed
- 39. Kosmider L, Sobczak A, Fik M, Knysak J, Zaciera M, Kurek J, Goniewicz ML. Carbonyl compounds in electronic cigarette vapors: effects of nicotine solvent and battery output voltage. Nicotine Tob Res. 2014;16(10):1319–26. CrossRef | PubMed
- 40. Farsalinos KE, Romagna G, Tsiapras D, Kyrzopoulos S, Voudris V. Evaluation of electronic cigarette use (vaping) topography and estimation of liquid consumption: implications for research protocol standards definition and for public health authorities’ regulation. Int J Environ Res Public Health. 2013;10(6):2500–14. CrossRef | PubMed
- 41. Marian C, O'Connor RJ, Djordjevic M, Rees VW, Hatsukami DK, Shields PG. Reconciling human smoking behavior and machine smoking patterns: implications for understanding smoking behavior and the impact on laboratory studies. Cancer Epidemiol Biomarkers Prev. 2009;18(12):3305–20. CrossRef | PubMed