Abstract
Background: Sydney has a large and highly mobile immigrant community. The pattern of paediatric tuberculosis (TB) disease in this highly cosmopolitan city is not well documented.
Methods: We reviewed data on all children notified with TB in New South Wales (NSW), Australia, from January 2014 to December 2015, complemented by an expanded dataset for children managed within the Sydney Children’s Hospitals Network (SCHN).
Results: Over the 2-year study period, 921 TB cases were identified in NSW, including 26 (2.8%) children younger than 15 years of age. Of 23 children and adolescents treated for TB in the SCHN, 21 (91.3%) had a history of recent immigration from, or travel to, a country with high TB incidence, and 7 (30.4%) reported contact with an infectious TB case in Australia. Fourteen (60.9%) children had microbiologically confirmed TB; of these, 5 (21.7%) had acid-fast bacilli on microscopy, 8 (34.8%) were positive by polymerase chain reaction and 11 (47.8%) were positive by culture. All Mycobacterium tuberculosis isolates were susceptible to first-line drugs. Ten (43.5%) cases were not vaccinated with bacille Calmette–Guérin (BCG), including all cases with severe disease: 2 with disseminated (miliary) TB and 3 with tuberculous meningitis.
Conclusion: Our findings emphasise the need for improved TB prevention and surveillance in children at high risk of exposure, particularly young children travelling to areas of high TB incidence.
Full text
Introduction
Tuberculosis (TB) remains a disease of major global public health significance, being the number one infectious disease killer on the planet.1,2 The World Health Organization (WHO) estimates that 1.8 million people died from TB in 20151,2, and autopsy studies suggest that TB is a common but frequently unrecognised cause of under-5 mortality in endemic areas.3 Recent modelling studies suggest that 240 000 children younger than 15 years of age died from TB in 2015, with 80% of these deaths occurring in children younger than 5 years of age.4
Australia has a low TB incidence (5–6 per 100 000 population per year), and the vast majority of cases occur in people born overseas.5,6 A 10-year review (2003–2012) identified 538 children who were treated for TB in Australia, accounting for nearly 5% of the national TB caseload – a proportion similar to that in other low-incidence settings.7 Studies in children of the natural history of disease indicated that the risk of developing active TB is greatest in the first 12 months following primary infection with Mycobacterium tuberculosis.6,8,9 Australian data indicate that 85% of TB cases in overseas-born children are diagnosed within 5 years of arrival in Australia, but these data do not capture recent overseas travel or bacille Calmette–Guérin (BCG) vaccination status.6
New South Wales (NSW) reports the highest number of TB cases of any Australian state or territory, with around 500 cases diagnosed per year.10 Given the size and cosmopolitan nature of its population, Sydney records the majority of TB cases in the state. The Sydney Children’s Hospitals Network (SCHN) comprises two hospitals: The Children’s Hospital at Westmead and Sydney Children’s Hospital, Randwick. Together, they provide tertiary/quaternary paediatric services to NSW. The disease spectrum and risk factors associated with TB in children diagnosed within the SCHN have not been previously described.
Methods
We obtained summary data on all TB cases reported to the NSW TB Program during a 2-year period (January 2014 to December 2015). The focus was on children 15 years of age or younger, which is the paediatric age cut-off used for national and global reporting. For more detailed information, we collected data on all children treated for TB within the SCHN during this period.
Data were collected in preparation for a national, opt-in child TB registry among children’s hospitals, as a partnership between the Australian and New Zealand Paediatric Infectious Diseases Group, the Paediatric Active Enhanced Disease Surveillance network, and the National Health and Medical Research Council Tuberculosis Centre of Research Excellence. Site-specific ethics approval was obtained to enter data from all children who commenced multidrug TB treatment into a secure database (WebSpirit – licensed to the Paediatric Trials Network Australia). Inclusion criteria included all children and adolescents aged 18 years or younger managed in the SCHN for TB disease, as either inpatients or outpatients. A variable age cut-off (usually 16–18 years) is applied when deciding on the appropriate timing of referral to adult services.
Following national TB guidance, we defined all countries with a TB incidence more than or equal to 40/100 000 population as ‘high TB incidence’ countries.11 Recent contact with an infectious TB case was defined as a household contact within the past 12 months. Children with TB were classified as ‘microbiologically confirmed’ or ‘clinically diagnosed’. Microbiological confirmation was established if the patient had a positive culture for M. tuberculosis complex, a positive polymerase chain reaction (PCR) test (including GeneXpert MTB/RIF) or visible acid-fast bacilli on sputum smear microscopy. Acid-fast bacilli seen on histology in the presence of caseating granulomas were also regarded as microbiological confirmation. One clinically diagnosed TB case did not meet criteria for bacteriological confirmation, but active TB was diagnosed by the clinical team, and the child received multidrug TB treatment.
All paediatric cases with TB disease involving the lung parenchyma, pleura or intrathoracic lymph nodes were classified as pulmonary disease, as in most paediatric case series. All other disease manifestations were classified as extrapulmonary. Children with both pulmonary and extrapulmonary disease manifestations were classified as having both. Confirmation of positive cultures, and routine drug susceptibility testing for isoniazid and rifampicin were conducted at the NSW Mycobacterium Reference Laboratory at the Institute of Clinical Pathology and Medical Research, Westmead. A successful outcome was defined as treatment completion with complete clinical recovery. Unsuccessful outcomes included death, treatment failure, loss to follow-up, or treatment completion with permanent sequelae.
Results
Table 1 summarises all TB cases reported in NSW in 2014 and 2015. In total, 921 TB cases were identified; 26 children (younger than 15 years of age) were notified, representing 2.8% of the total case load. Over the 2-year study period, doctors in the SCHN managed 69.2% (18/26) of all children (younger than 15 years of age) diagnosed with TB in NSW. An additional 5 adolescents (aged 15–16 years) were managed in the SCHN, giving a total of 23 children and adolescents: 6 at Sydney Children’s Hospital, Randwick, and 17 at The Children’s Hospital at Westmead. Ages of TB cases managed in the SCHN ranged from 5 weeks to 15 years, with a median of 9 years.
Table 1. Overview of TB cases diagnosed in NSW over a 2-year period (2014–2015)a
| Variable | Year of diagnosis | Total n (%) |
||
| 2014 | 2015 | |||
| Age group (years) | ||||
| 0–4 | 5 | 9 | 14 (1.5) | |
| 5–9 | 4 | 0 | 4 (0.4) | |
| 10–14 | 3 | 5 | 8 (0.9) | |
| 15–19 | 12 | 16 | 28 (3.0) | |
| 20–39 | 221 | 202 | 423 (45.9) | |
| 40–59 | 103 | 98 | 201 (21.8) | |
| 60–79 | 85 | 87 | 172 (18.7) | |
| 80+ | 42 | 29 | 71 (7.7) | |
| Total | 475 | 446 | 921 (100) | |
| Sex | ||||
| Male | 239 | 246 | 485 (52.7) | |
| Female | 236 | 200 | 436 (47.3) | |
| Identification method | ||||
| Clinical | 95 | 95 | 190 (20.6) | |
| Laboratory confirmed | 380 | 351 | 731 (79.4) | |
| Disease site | ||||
| PTB only | 264 | 253 | 517 (56.1) | |
| EPTB only | 172 | 161 | 333 (36.2) | |
| PTB + EPTB | 39 | 32 | 71 (7.7) | |
| Disease classification | ||||
| New active | 451 | 420 | 871 (94.6) | |
| Recurrent | 24 | 22 | 46 (5.0) | |
| Unknown | 0 | 4 | 4 (0.4) | |
| Indigenous status | ||||
| Australian born | Indigenousb | 8 | 3 | 11 (9.9) |
| Non-Indigenous | 54 | 46 | 100 (90.1) | |
| Not stated/unknown | 0 | 0 | 0 (0.0) | |
| Total | 62 | 49 | 111 (12.1) | |
| Overseas born | Indigenousb | 1 | 0 | 1 (0.1) |
| Non-Indigenous | 411 | 397 | 808 (99.8) | |
| Not stated/unknown | 1 | 0 | 1 (0.1) | |
| Total | 413 | 397 | 810 (87.9) | |
| HIV infection status | ||||
| Test result negative | 344 | 326 | 670 (72.7) | |
| Test result positive | 6 | 3 | 9 (1.0) | |
| Test result not known | 3 | 8 | 11 (1.2) | |
| Test not done | 122 | 109 | 231 (25.1) | |
EPTB = extrapulmonary TB; HIV = human immunodeficiency virus; PTB = pulmonary TB; TB = tuberculosis
a Data sourced from the NSW Notifiable Conditions Information Management System
b Aboriginal and/or Torres Strait Islander
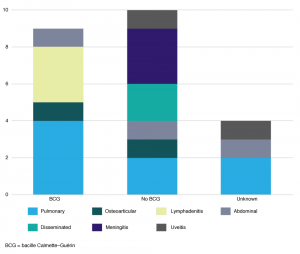
Table 2 reflects the demographic profile and risk factors identified in children diagnosed with TB in the SCHN. Recent migration from (10; 43.5%), or visit to (11; 47.8 %), a high TB incidence country were leading risk factors. The most frequent travel destination was India (6/11; 54.5%); other countries listed were Vietnam, China, Bangladesh, Sri Lanka and Tonga. Where travel duration was documented (in eight cases), it ranged from 1 to 3 months. Other risk factors included recent close contact with an infectious TB patient (7; 30.4%), and Australian-born children of parents from a high TB incidence country (22; 95.7%). None of the children included in our series identified as Aboriginal or Torres Strait Islander. Ten (43.5%) of the children had not received BCG vaccination, including all children treated for severe forms of TB: two with disseminated (miliary) TB and three with tuberculous meningitis (Figure 1). Three of these severe cases reported travel for 1–3 months to a TB-endemic country, whereas two had no history of travel or recent immigration. Two (40%) of the children with severe TB were more than 5 years old. None of the children were HIV infected, received immunosuppressive therapy or were known to have an underlying immunodeficiency.
Table 2. Demographics and risk factors in children treated for TB in the SCHN
| Demographics (N = 23) | n (%) |
| Age group (years) | |
| <2 | 4 (17.4) |
| 2–4 | 4 (17.4) |
| 5–9 | 4 (17.4) |
| 10–14 | 6 (26.1) |
| 15–18 | 5 (21.7) |
| Sex | |
| Male | 16 (69.6) |
| Female | 7 (30.4) |
| Risk factors identified (N = 23) | |
| Born in high TB incidence country | 10 (43.5) |
| At least one parent born in high TB incidence country | 22 (95.7) |
| Refugee | 3 (13.0) |
| Recenta overseas travelb (high TB incidence country) | 11 (47.8) |
| Recenta closec or household TB contact | 7 (30.4) |
| No BCG vaccination | 10 (43.5) |
| Immunosuppressiond | 0 (0) |
BCG = Bacille Calmette–Guérin; HIV = human immunodeficiency virus; SCHN = Sydney Children’s Hospitals Network; TB = tuberculosis
a Within the past 12 months
b Overseas travel to the following countries: India (6/11), Vietnam, China, Bangladesh, Sri Lanka or Tonga
c Exposed to an adult with known pulmonary TB for extended period outside the household
d HIV infected, use of high-dose steroids or other immunosuppressive medication, known underlying immunodeficiency.
Figure 1. Number of children with tuberculosis by site of disease and BCG vaccination status (click to enlarge)
Fourteen (60.9%) cases were microbiologically confirmed, and 9 (39.1%) were diagnosed on clinical features only. Of those microbiologically confirmed, 5 (21.7%) had acid-fast bacilli seen on microscopy, 8 (34.8%) were positive by polymerase chain reaction and 11 (47.8%) were positive by culture. Table 3 shows the disease spectrum observed in the SCHN TB cases, stratified by age group. Of the 16 patients with pulmonary TB (with or without extrapulmonary involvement), five were positive on sputum culture, of whom one was positive on sputum acid-fast bacilli smear. A further four had microbiological confirmation from a specimen other than sputum: two from bronchoalveolar lavage, one from cerebrospinal fluid and one from pleural biopsy. Of the seven children with extrapulmonary TB, four had tissue sent for culture; none had a positive TB culture, but one axillary lymph node biopsy was PCR positive. All cultured isolates were fully susceptible to first-line tuberculous drugs via conventional phenotypic testing.
Table 3. Disease spectrum in children treated for TB in the SCHN
| Disease manifestation | Age group (years) | ||||
| <2 | 2–4 | 5–10 | 11–18 | Total | |
| PTB only | 0 | 1 | 2 | 5 | 8 (34.8%) |
| Uncomplicated lymph node disease | 0 | 0 | 0 | 0 | 0 |
| Complicated lymph node disease | 0 | 1 | 2 | 1 | 4 |
| Adult-type cavitary disease | 0 | 0 | 0 | 2 | 2 |
| Pleural disease | 0 | 0 | 0 | 2 | 2 |
| EPTB only | 1 | 3 | 0 | 3 | 7 (30.4%) |
| Peripheral lymphadenitis | 1 | 1 | 0 | 0 | 2 |
| Abdominal TB | 0 | 1 | 0 | 0 | 1 |
| Osteoarticular TB | 0 | 1 | 0 | 1 | 2 |
| TB uveitis | 0 | 0 | 0 | 2 | 2 |
| PTB + EPTB | 3 | 0 | 2 | 3 | 8 (34.8%) |
| Disseminated (miliary) disease | 1 | 0 | 1 | 0 | 2 |
| TBM + PTB | 2 | 0 | 0 | 1 | 3 |
| Other | 0 | 0 | 1 | 2 | 3 |
| Total | 4 (17.4%) | 4 (17.4%) | 4 (17.4%) | 11 (47.8%) | 23 (100.0%) |
EPTB = extrapulmonary TB; PTB = pulmonary TB; SCHN = Sydney Children’s Hospitals Network; TB = tuberculosis; TBM = tuberculous meningitis
Of those positive by GeneXpert MTB/RIF, none had an rpoB mutation suggestive of rifampicin resistance. Among children with peripheral tuberculous lymphadenitis, two had axillary lymphadenitis (both younger than 2 years of age) and one had cervical lymphadenitis (11 years of age). Both children with axillary lymphadenitis had received BCG vaccination, but it was impossible to determine from the available records whether the involved lymph nodes were on the same side as the vaccination scar. Two patients had osteoarticular TB: a 2-year-old boy with disseminated osteomyelitis and a 15-year-old boy with L4–L5 discitis and osteomyelitis.
All children received multidrug regimens given as directly observed therapy (DOT), supervised by the local TB (‘Chest’) clinic, as per WHO recommendations. Three patients developed paradoxical reactions: two with tuberculous meningitis and one with worsening cervical lymphadenitis 2–3 months after commencing TB treatment. The two patients with tuberculous meningitis who developed paradoxical reactions did so when steroids given during the first month of TB treatment were weaned; this required prolonged steroid courses with slow tapering. Most children (22; 95.7%) completed their TB treatment and had an excellent clinical outcome.
Only one Australian-born boy, diagnosed with possible tuberculous uveitis, had apparent treatment failure. He had a positive gamma-interferon assay, but normal chest X-ray and no other signs or risk factors for TB; the TB diagnosis in this case remains highly uncertain. Three patients were treated for tuberculous meningitis. An 11-month-old baby developed right-sided hemiplegia and shunt-dependent hydrocephalus, but the other two children (18 months and 14 years of age) had no neurological sequelae on follow-up. A 6-week-old baby developed haemophagocytic lymphohistiocytosis as a result of disseminated congenital TB disease, which required chemotherapy and 12 months of TB treatment. On follow-up at 2 years of age, he had normal development and was not on any immunosuppressing medication.
Discussion
Childhood TB is an uncommon disease in Australia5, but ongoing vigilance is required because children occasionally develop severe disease manifestations whose treatment outcomes are highly dependent on early appropriate treatment. Furthermore, children are important epidemiological ‘markers’ of TB transmission in communities. Our data reflect the broad spectrum of TB disease observed in settings where the majority of TB cases are imported disease. More than 90% of children treated for active TB in our study had a history of recent immigration from, or travel to, a high TB incidence country. None of the children with severe disseminated disease had received the BCG vaccine.
Regarding the disease spectrum observed, children younger than 2 years of age were more likely than older children to have disseminated disease including tuberculous meningitis. Adult-type cavitating lung disease and pleural effusion occurred only in children older than 10 years of age, as described in the literature.13 Two infants treated for axillary lymphadenitis received BCG vaccination after birth. We note that in one, microbiological confirmation was with GeneXpert MTB/RIF, which does not differentiate M. bovis BCG from other species in the M. tuberculosis complex. We cannot exclude the possibility of misdiagnosis and that these children might have had BCG adenitis with unnecessary treatment for TB.9
International travel and migration are important contributors to the TB caseload in Australia12, and recent immigrants from high TB incidence countries account for the vast majority of cases across the age spectrum.7,13,14 This is reflected in our series from NSW, where the most frequent risk factors among children treated for TB were having parents born in a high TB incidence country, and a history of recent immigration from, or travel to, high TB incidence countries. This contrasts with data reported from Queensland, Australia, where more than 50% of children treated for TB were cross-border patients from Papua New Guinea.15 Australian Bureau of Statistics data reveal that international travel to and from Australia has increased considerably in the past 10 years – the number of Australian residents departing for trips abroad in 2014–15 was nearly 10 million, almost double the figure recorded for 2004–05.16 The main destination countries in 2015 were Indonesia, Thailand, India and China, which are all considered high TB incidence countries. To achieve the strategic goal of TB elimination in Australia, key priorities include identifying TB infection in recently arrived migrants (including those with refugee and asylum-seeker backgrounds) before disease progression, and preventing TB in children travelling to high TB incidence countries.16
The NSW TB Program focuses on case finding, early diagnosis and effective treatment to minimise and eliminate local M. tuberculosis transmission. NSW has a strong surveillance system in place, whereby all patients diagnosed with TB are notified to public health units in accordance with the NSW Public Health Act 2010.10 Maintaining a low incidence of TB despite the large-scale immigration from TB-endemic countries is highly dependent on effective pre-migration screening.17 It is recommended that all migrants, including children, be screened for TB and treated before migration, if required.6 New pre-migration TB screening protocols for children were introduced at the end of 2015, involving an interferon-gamma release assay (IGRA) test or tuberculin skin test (TST) in children aged 2–10 years, followed by a chest X-ray in those who test positive.6,7 Post-arrival screening of refugee and asylum-seeker children is also recommended nationally to identify those missed or not eligible for pre-migration screening.6 In those without active TB, initiation of treatment of latent TB infection in children aged younger than 5 years is the standard of care, but the use of chemoprophylaxis for latent TB infection remains highly variable in those aged 5 years or older.6
Another key TB prevention strategy for children is vaccination of those travelling to high TB incidence countries. BCG vaccination reduces the risk of M. tuberculosis infection and disease progression, with more than 70% efficacy against severe forms of disease in young children.18,19 BCG is not included in the routine vaccination schedule in Australia, but should be considered when vulnerable children (e.g. aged younger than 5 years) will be exposed to a high-risk environment, such as travelling to their parent’s country of origin.20 All patients with severe disease (5/5) in this study were born in Australia and had not received the BCG vaccine, despite their parents originating from high TB incidence countries. Efforts to increase awareness of BCG vaccination, especially in parents of young children who visit friends and relatives in their home countries, should be encouraged, and ongoing supply of the BCG vaccine and ease of vaccination access should be ensured.21 BCG vaccination should be actively promoted for these children.22 Although false-positive TST results are possible in those who are BCG vaccinated, the IGRA test still provides a specific result in people who have received BCG vaccination.23
Given the relative infrequency of childhood TB, and the complexities of diagnosis and management, we have initiated a multidisciplinary meeting within the SCHN for detailed case review. The meetings include infectious diseases physicians, microbiologists, TB nurses, pharmacists, and delegates from the state TB program and local TB clinics. We anticipate that this paediatric TB multidisciplinary team will improve coordination of care for individual patients and support ongoing policy development in child TB prevention and management.
It should be recognised that our study cohort was relatively small, and we missed children who did not require hospitalisation, which might bias our cohort towards more severe disease. Nonetheless, our cohort included more than two-thirds of all child TB cases managed in NSW during the study period and therefore provides a realistic overview of the TB disease spectrum observed in a tertiary referral network in Australia. A future comprehensive review of child TB cases throughout Australia and New Zealand would address some of these limitations.
Conclusion
Children suffer a considerable and increasingly recognised disease burden in high TB incidence countries, from which Australia continues to receive migrants and refugees. Disease rates in Australia remain very low, with minimal local transmission, but recent migrants frequently return to their country of origin, potentially leading to infection of children born in Australia. In pursuing the strategic goal of TB elimination in Australia, several challenges remain, including reducing the large pool of latent infection among immigrants, dealing with the risk of infection and re-infection during travel to high TB incidence settings, and educating parents about the benefits of BCG vaccination in young children likely to travel to TB-endemic countries.
Acknowledgements
We thank the neurologists, pulmonologists, physicians, microbiologists and TB nurses involved in the management of the patients described in this study, for their contributions and exemplary clinical care. We also acknowledge the ANZ TB database investigator group that includes Kristine Macartney, Nigel Curtis, Asha Bowen, Celia Cooper, Lesley Voss, Eugene Athan and Joshua Francis.
Peer review and provenance
Externally peer reviewed, not commissioned.
Copyright:
© 2018 Al Yazidi et al. This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International Licence, which allows others to redistribute, adapt and share this work non-commercially provided they attribute the work and any adapted version of it is distributed under the same Creative Commons licence terms.
References
- 1. World Health Organization. The end TB strategy 2015. Geneva: WHO; 2015 [cited 2017 Oct 15]. Available from: www.who.int/tb/post2015_strategy/en/
- 2. World Health Organization. Global tuberculosis report 2016. Geneva: WHO; 2016 [cited 2017 Sep 1]. Available from: apps.who.int/medicinedocs/en/d/Js23098en/
- 3. Graham SM, Sismanidis C, Menzies HJ, Marais BJ, Detjen AK, Black RE. Importance of tuberculosis control to address child survival. Lancet. 2014;383(9928):1605–7. CrossRef | PubMed
- 4. Dodd PJ, Yuen CM, Sismanidis C, Seddon JA, Jenkins HE. The global burden of tuberculosis mortality in children: a mathematical modelling study. Lancet Glob Health. 2017;5(9):e898–906. CrossRef | PubMed
- 5. Toms C, Stapledon R, Waring J, Douglas P; National Tuberculosis Advisory Committee, for the Communicable Diseases Network Australia, and the Australian Mycobacterium Reference Laboratory Network. Tuberculosis notifications in Australia, 2012 and 2013. Commun Dis Intell Q Rep. 2015;39(2):E27–35. PubMed
- 6. Denholm J, Bailes M, Francis J. Chapter 1: Tuberculosis. In: Chaves NJ, Paxton G, Biggs BA, Thambiran A, Smith M, Gardiner J, et al. Recommendations for comprehensive post-arrival health assessment for people from refugee-like backgrounds. Sydney: ASID; 2016 [cited 2017 Aug 7]. Available from: www.asid.net.au/documents/item/1225
- 7. Teo SS, Tay EL, Douglas P, Krause VL, Graham SM. The epidemiology of tuberculosis in children in Australia, 2003–2012. Med J Aust. 2015;203(11):440. CrossRef | PubMed
- 8. Marais BJ, Gie R, Schaaf H, Hesseling AC, Obihara CC, Starke JJ, et al. The natural history of childhood intra-thoracic tuberculosis: a critical review of literature from the pre-chemotherapy era. Int J Tuberc Lung Dis. 2004;8:392–402. PubMed
- 9. Perez-Velez CM, Marais BJ. Tuberculosis in children. N Engl J Med. 2012;367(4):348–61. CrossRef | PubMed
- 10. NSW Heath. Tuberculosis New South Wales: 2012–2014. Sydney: NSW Health [cited 2017 Aug 7]. Available from: www.health.nsw.gov.au/Infectious/tuberculosis/Documents/tb-report-2012-14.pdf
- 11. NSW Health. List of Countries with a Tuberculosis Incidence of 40 cases per 100,000 Persons or Greater. Sydney: State of New South Wales NSW Ministry of Health; 2018 [cited 2018 Aug 27]. Available from: www.health.nsw.gov.au/infectious/tuberculosis/documents/countries-incidence.pdf
- 12. Britton PN, Perez-Velez CM, Marais BJ. Diagnosis, treatment and prevention of tuberculosis in children. NSW Public Health Bull. 2013;24:15–21. CrossRef | PubMed
- 13. McBryde ES, Denholm JT. Risk of active tuberculosis in immigrants: effects of age, region of origin and time since arrival in a low-exposure setting. Med J Aust. 2012;197(8):458–61. CrossRef | PubMed
- 14. Britton PN, Yeung V, Lowbridge C, Isaacs D, Marais BJ. Spectrum of disease in children treated for tuberculosis at a tertiary children's hospital in Australia. J Pediatric Infect Dis Soc. 2013;2(3):224–31. CrossRef | PubMed
- 15. Donnan EJ, Coulter C, Simpson G, Clark J, Nourse C. Paediatric tuberculosis in Queensland, Australia: overrepresentation of cross-border and Indigenous children. Int J Tuberc Lung Dis. 2017;21(3):263–9. CrossRef | PubMed
- 16. Australian Bureau of Statistics. Canberra: ABS; 2018. Migration, Australia 2015–16; 2017 March 30 [cited 2017 June 29]; [about 2 screens]. Available from: www.abs.gov.au/ausstats/[email protected]/mf/3412.0
- 17. Australian Government Department of Health. Canberra: Department of Health; 2018. National Tuberculosis Advisory Committee. The strategic plan for control of tuberculosis in Australia: 2011–2015. Canberra: NTAC; 2014 [cited 2017 Jun 29]; [about 12 screens]. Available from: www.health.gov.au/internet/main/publishing.nsf/content/cda-cdi3603i.htm
- 18. Roy A, Eisenhut M, Harris RJ, Rodrigues LC, Sridhar S, Habermann S, et al. Effect of BCG vaccination against Mycobacterium tuberculosis infection in children: systematic review and meta-analysis. BMJ. 2014;349:g4643. CrossRef | PubMed
- 19. Trunz BB, Fine P, Dye C. Effect of BCG vaccination on childhood meningitis and miliary tuberculosis worldwide: a meta-analysis and assessment of cost-effectiveness. Lancet. 2006;367(9517):1173–80. CrossRef | PubMed
- 20. Australian Government Department of Health. The Australian Immunisation Handbook. 10th ed. Canberra: Department of Health; 2017 [cited 2017 Jul 1]. Available from: immunise.health.gov.au/internet/immunise/publishing.nsf/Content/Handbook10-home~handbook10part4~handbook10-4-20
- 21. Marais BJ, Seddon JA, Detjen AK, van der Werf MJ, Grzemska M, Hesseling AC, et al. on behalf of the WHO Child TB sub-group. Interrupted BCG vaccination is a major threat to global child health. Lancet Resp Med. 2016;4:251–3. CrossRef | PubMed
- 22. Gwee A, Rodrigo R, Casalaz D, Ritz N, Curtis N. Infants born in Australia to mothers from countries with a high prevalence of tuberculosis: to BCG or not to BCG? Med J Aust. 2013;199(5):324–6. CrossRef | PubMed
- 23. Pai M, Denkinger CM, Kik SV, Rangaka MX, Zwerling A, Oxlade O, et al. Gamma interferon release assays for detection of Mycobacterium tuberculosis infection. Clin Microbiol Rev. 2014;27(1):3–20. CrossRef | PubMed